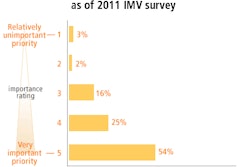
Radiopharmaceutical developer Ion Beam Applications (IBA) and drug company Wilex have completed a follow-up meeting with the U.S. Food and Drug Administration (FDA) to discuss the next steps in the development process for their Redectane PET/CT radiopharmaceutical.
Being developed for presurgical diagnosis of clear-cell renal cell cancer (ccRCC), Redectane is the radiolabeled form of the antibody girentuximab, which targets ccRCC and accumulates in tumor tissue. It can then be visualized during PET imaging.
During a recent meeting, the FDA offered the option of discussing the regulatory pathway, timing, and design of a second study for Redectane with an FDA advisory committee. Wilex accepted the offer, and the FDA is expected to confirm the advisory committee discussion after internal verification of meeting logistics, according to the companies.
In related developments, Wilex's manufacturing partner Avid Bioservices has successfully completed the production run of the third consecutive consistency lot for process validation of girentuximab. IBA, which is responsible for radioactive labeling of the antibody, is now assembling commercial production data.
The new facility for central manufacturing and subsequent quality assurance of Redectane has been completed, and IBA is preparing the documentation, according to the firms.