C-Ray Therapeutics has received active status from the U.S. Food and Drug Administration (FDA) for its Type II Drug Master File (DMF) for copper-64 (Cu-64) chloride for radiolabeling.
The file is listed under DMF No. 43568 and is now available for reference by radiopharmaceutical developers in support of investigational new drug and new drug application submissions.
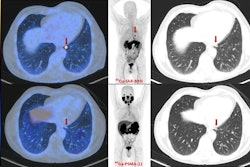
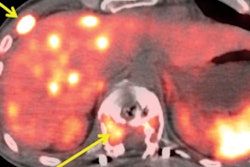
Cu-64 offers a half-life of about 12.7 hours, significantly longer than gallium-68 (68 minutes) and fluorine-18 (110 minutes). This allows for centralized production, shelf lives of up to 48 hours, and reduced scheduling pressure at imaging centers, according to the company. A 2026 study published in European Urology evaluating Cu-64 SAR-bisPSMA in prostate cancer patients found the agent detected more than twice as many lesions as Ga-68 PSMA-11, with a positive detection rate of 78% versus 36%, C-Ray said.
C-Ray's manufacturing process includes an in-house nickel-64 recycling system with a recovery rate of up to 90%, enabling multibatch operation and reduced raw material costs. Current batch capacity exceeds 2 Ci at end of bombardment, the firm said.