A new double-contrast MRI technique in development could help the modality distinguish very small tumors from normal tissue, according to a study published May 25 in Nature Nanotechnology.
The technique shows potential for even more effective diagnosis of cancers and other diseases, wrote a team led by senior author Dr. Yuanpei Li of the University of California, Davis.
"[The platform we designed] makes MRI much more sensitive and selective than the conventional techniques used in cancer diagnosis," the group wrote.
The technique is based on a phenomenon called MR tuning -- reactions that occur between nanoscale magnetic elements -- and takes advantage of chemical probes that produce signals on MRI, according to the researchers. One reaction enhances the signal, and the other "quenches" it, the university said in a statement.
"Previous studies have shown that quenching depends on the distance between the magnetic elements," it said. "This opens new possibilities for noninvasive and sensitive investigation of a variety of biological processes by MRI."
Li and colleagues developed a contrast agent that generates two MR signals that "suppress each other until they reach the target, at which point they both increase contrast between a tumor and surrounding tissue," the university said in a statement. The agent consists of two parts: nanoparticles of superparamagnetic iron oxide (SPIO) and pheophorbide a-paramagnetic manganese (P-Mn), combined in a lipid envelope. The particles emit strong signals on MRI, but when they are close together, they cancel each other out; when they enter tumor tissue, however, both signals reappear.
The researchers call this process "two-way MR tuning (TMRET)" and combined it with imaging analysis software that gathered data from the agent and reconstructed images in a process called double-contrast enhanced subtraction imaging. In an animal study, Li's team found that their agent produced a tumor signal 10 times as strong as normal tissue.
Li's group plans to continue their research on use of the probe before applying for an investigational new drug approval from the U.S. Food and Drug Administration (FDA).
"[This technique is] a significant advance," Li said in the university's statement. "It could help detect very small early-stage tumors."



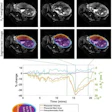
![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=100&q=70&w=100)





![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)