Performing MRI scans before biopsy on men with high prostate-specific antigen (PSA) scores could reduce prostate cancer death rates by as much as 18% versus no screening, while also reducing the harms of screening compared with other strategies, according to a March 11 study in JAMA Network Open.
The findings could help clinicians better tailor prostate cancer screening based on risk of developing prostate cancer, rather than simply screening all men based on their age, researchers from University College London said in a statement released by the university.
"The scenario generating the most benefits would be to screen men with a 3.5% risk of getting prostate cancer over the next 10 years -- that is, roughly half of all men aged 55 to 69," a team led by Dr. Thomas Callender said. "Such a program could prevent up to 16% of prostate cancer deaths ... [and] would also be more cost-effective than screening all men aged 55 to 69."
Although prostate cancer is a leading cause of death from cancer among men, "there is no screening program [in the U.K.] because the harms of screening are considered to outweigh the benefits," Callender said.
But men at higher genetic risk of prostate cancer are more likely to benefit from screening and less likely to be harmed, he noted. And multiparametric MRI could potentially help mitigate the harms of biopsy by triaging for biopsy only those men with clinically suspicious disease.
Callender's group sought to investigate whether they could develop a prostate cancer screening strategy that could save lives while reducing the harms of screening. They developed an MRI-first prostate cancer screening protocol -- with MRI performed after PSA screening and before biopsy -- that they postulated would be more effective than a biopsy-first protocol via a study that used a decision model created from a hypothetical cohort of 4.5 million English men between 55 and 69.
The team created a benefit-to-harms profile that considered potential deaths from prostate cancer, quality-adjusted life-years, overdiagnosis, and biopsies, as well as costs associated with each strategy within a 10-year risk time frame. They analyzed the deaths from prostate cancer with three strategies:
- No screening
- MRI before biopsy based on age
- MRI before biopsy based on prostate cancer risk level, ranging from 2% to 10%
The investigators found that age-based screening using MRI before biopsy for prostate cancer saved the most lives in the study: Deaths were reduced by 18% for men with the MRI-first protocol and age-based screening compared with no screening.
But basing who got MRI scans on the risk level of the men rather than age produced a conundrum -- the number of harms related to screening was lower, but the strategy did not save as many lives as the age-based protocol.
| Impact of MRI-first prostate cancer screening on outcomes based on risk level | ||
| Measure | MRI-first screening at 2% risk of cancer | MRI-first screening at 10% risk of cancer |
| No. of overdiagnosed cancers | -10.4% | -72.6% |
| No. of MRI scans overall | -21.7% | -53.5% |
| No. of biopsies | -22% | -53.4% |
| Deaths from prostate cancer compared with no screening | -18% | -7% |
| Deaths from prostate cancer compared with age-based screening | 1.6% | 13.3% |
As for cost, the team discovered that the most effective screening strategy at "willingness to pay" thresholds (that is, the value a health system would estimate is appropriate to pay for one year of full health in a patient) of $26,000 and $39,000 per quality-adjusted life-year gained was MRI-first risk-based screening at 10-year absolute risk thresholds of 8.5% and 7.5%.
Callender and colleagues found that the most effective screening scenario would be to screen men with a 3.5% risk of getting prostate cancer over the next 10 years -- a protocol that could prevent up to 16% of prostate cancer deaths and reduce overdiagnosis by 27%, the university said. Screening men at this threshold (3.5%) would also be more cost-effective than screening all men ages 55 to 69.
The study highlights MRI's promise for reducing the harms of prostate cancer screening, co-author Dr. Mark Emberton concluded.
"Our study shows that screening for prostate cancer -- which could save between 16% and 20% of prostate cancer deaths -- might be possible with targeted screening using genetic risk and MRI as part of the diagnostic pathway," he said in the university statement. "This paves the way for further clinical trials to study the real-world implementation of such a screening program."




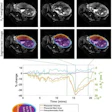
![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=100&q=70&w=100)



![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)