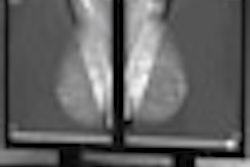
(Booth 227) Hologic plans to feature its work-in-progress breast tomosynthesis device, Selenia Dimensions, a 3D full-field version of digital mammography that may make it possible to see cancers even earlier, according to the Bedford, MA-based company.
The company also will display its automated breast density assessment tool, Quantra, recently cleared by the U.S. Food and Drug Administration (FDA). Quantra is a software application for use with Hologic's digital mammography systems that creates an internal 3D model of the breast from which it estimates fibroglandular tissue volume and the total breast volume.

The company also will highlight its MammoSite radiation therapy system, a balloon catheter that can be inserted into the cavity created by a lumpectomy to deliver radiation from inside the cavity over the course of five days.
For breast biopsy, Hologic will showcase its upright StereoLoc II system, as well as its ATEC (automatic tissue excision and collection) device. Also in its booth will be Celero, a vacuum-assisted spring-loaded core biopsy device for breast ultrasound, which recently received the CE Mark. In addition, SecurMark is a family of biopsy site identifiers designed for Celero and ATEC.
As for bone health, Hologic has received clearance from the FDA to incorporate the World Health Organization's fracture risk calculator, FRAX, into its bone densitometer systems. FRAX helps identify patients who are at high risk of fracture but who would not be candidates for preventive therapy according to the traditional T-score, the standard for diagnosing osteoporosis.
Hologic also will feature MammoPad radiolucent breast cushions on its Selenia systems. MammoPad is designed to reduce women's discomfort during mammography exams and ensure optimal breast positioning.
In computer-aided detection (CAD), Hologic will talk up recent published clinical papers that highlight the utility of CAD and the company's R2 software in improving the accuracy of screening mammography.
One study, in the April 2008 issue of the American Journal of Roentgenology, used R2 technology as part of a large-scale breast screening trial that compared a single radiologist using CAD to double reading without CAD. The study compared the recall rate, sensitivity, positive predictive value, and cancer detection rate for single reading with CAD versus double reading without CAD, and found that R2 CAD increases sensitivity in cancer detection with only a small increase in recall rate, according to the company.
In the second and perhaps even more influential study, published this month in the New England Journal of Medicine, U.K. researchers used R2 software in a multicenter trial comparing the cancer detection rates of screening with single reading and CAD to that of double reading. The group found that single reading with CAD produced a sensitivity of 87.2%, while double reading recorded a sensitivity of 87.7%. Single reading with CAD had a marginally higher recall rate of 3.9%, versus 3.4% for double reading.
Finally, the company will continue the Digital Mammography Training and Self-Assessment workshop it debuted at the 2005 RSNA meeting.