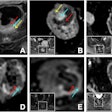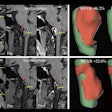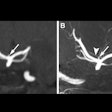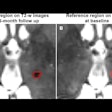
Polarean Imaging is touting the results of two phase III clinical trials for its drug-device combination that uses hyperpolarized xenon-129 (Xe-129) gas with MRI to visualize and quantify regional lung function.
When polarized with Polarean's system, Xe-129 enables functional, regional, and quantitative MR imaging of the lungs. Patients inhale Xe-129 as a gas during a 10-second breath-hold procedure. During clinical trials, ventilation in zones of interest was quantified and compared with images, similarly quantified, from a different imaging modality.
Both clinical trials met their primary end points within the prospectively defined equivalence margin (± 14.7%) when compared with a reference standard approved by the U.S. Food and Drug Administration (FDA), 133-xenon scintigraphy imaging.
Polarean plans to submit a new drug application (NDA) to the FDA in the third quarter of this year.