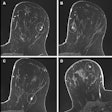
An advisory panel of the U.S. Food and Drug Administration today unanimously approved a premarket approval (PMA) application from Hologic for a digital breast tomosynthesis (DBT) system. If the FDA signs off on the final PMA, Hologic will be the first vendor to sell a DBT system on the U.S. market.
FDA panel gives nod to Hologic tomosynthesis PMA
Sep 23, 2010
Latest in Breast
Solis Mammography expands into Missouri
October 15, 2025
qHDMI differentiates radial scars from invasive breast cancer
October 15, 2025