Adrenergic Nervous System of the Heart
Physiology & Pharmacology
The sympathetic and parasympathetic innervation of the heart plays a major role in the regulation of cardiac function [14]. Adrenergic fibers that innervate the heart originate in the left and right stellate ganglia [9]. The left stellate innervates the right ventricle, whereas the right stellate innervates the anterior and lateral portions of the heart. The adrenergic fibers travel in the subendocardium following the coronary vessels [9]. At a cardiac level, sympathetic activation results in an increased heart rate (chronotropic effect), augmented contractility (inotropic effect), and enhanced atrioventricular conduction [9].
Sympathetic nervous system dysfunction plays a role in heart failure [18]. Both increased neuronal release of norepinephrine and decreased efficiency of NE uptake contribute to increased cardiac adrenergic drive [18]. Significant reduction in mortality in heart failure patients can be achieved with the use of beta- and alpha-adrenoreceptor blocking drugs [18].
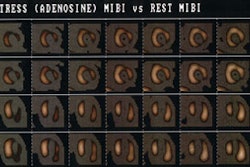
Metaiodobenzylguanidine (MIBG) is a guanethidine analog that mimics the neuronal transport and storage of norepinephrine [26]. I-123 MIBG has been used to study the cardiac sympathetic nervous system because of its high cardiac uptake. Non-neuronal activity clears rapidly from the heart between 15 and 60 minutes. In the heart, MIBG is taken up and store by the postganglionic, presynaptic nerve endings [21,29]. There are two mechanisms for MIBG uptake in adrenergic tissues: a type I neuronal uptake system and a extraneuronal type 2 uptake system (a minor role in uptake related to a carrier-facilitated process and diffusion into the myocytes) [26,29]. As with norepinephrine, I-123 MIBG uptake is predominantly mediated (70%) via the ATP energy dependent, type I uptake mechanism [9,26]. After depolarization, MIBG is released into the synaptic cleft, like norepinephrine, but it is not further metabolized [21]. MIBG stored within the neuron is also not further metabolized (it is not a substrate for monoamine oxidase [26]) and only about 10% of the dose is altered after several days, primarily by deiodination. The agent produces no measurable pharmacologic effects. Uptake of I-123 is usually homogeneous within the myocardium, although uptake may become more heterogeneous or decrease within the inferior (men) or lateral (woman) wall as the patient ages [1,2]. Tracer washout also increases with increasing age [2].
11C-meta-hydroxy-ephedrine (HED) is a PET agent that can be used for neuronal imaging [33]. The agent has higher uptake selectivity compared to MIBG and is better in differntiating between innervated and denervated myocardium [33].
Technique:
Patients should fast for 6 hours prior to the exam [13]. Thyroid uptake is blocked by the oral administration of 500 mg potassium perchlorate (Lugols solution) or a saturated solution of potassium iodide given 30 minutes before injection of the radiotracer [20]. Other authors recommend giving 1mg of potassium iodide from one day before to 1 day after the exam to block thyroid uptake [30]. Patients are injected at rest [33]. Medications that might interfere with catecholamine uptake, such as various antidepressants, antipsychotics, and some calcium channel blockers should be held for 24 hours prior to adminsitration of the tracer [33]. The dose used for the exam is 3 to 10 mCi of I-123 MIBG given over 1 minute (the higher dose may be required for patients with severe cardiac dysfunction if SPECT images are to be obtained) [8,9,13,33].
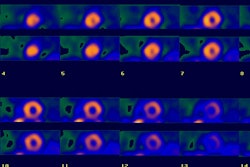
Early (10-30 minute post injection) and delayed (4 hour) planar and SPECT imaging is performed. The neuronal accumulation of MIBG reaches its maximum at 4 hours after injection (hence the delayed image represents actual neuronal uptake as opposed to interstitial uptake on the early images) [26,33]. A low-energy parallel hole collimator is used for imaging [20], however, other authors recommend using a medium energy or I-123 collimator in order to minimize noise from scatter [35]. A 20% window is used and centered over the 159 keV I-123 photon peak [9,20]. An anterior planar image of the chest is acquired for 5 to 10 minutes prior to initiation of SPECT imaging using a 128x128 matrix (some centers also acquire a planar LAO image [9]). Planar images are limited by superimposition of thoracic structures that can also demonstrate MIBG uptake (such as the luns and liver) as well as superimposition of different myocardial segments [29]. Tomographic images are performed to overcome these limitations [29]. SPECT images are obtained every 6 degrees for 30-40 seconds in a 64x64 matrix for a 180 degree rotation (RAO to LPO). Unfortunately, if global myocardial uptake of MIBG is severely reduced, it can be difficult to acquire tomographic images of sufficient quality [29].
Findings:
In healthy subjects, MIBG uptake is slightly lower in the inferior wall (likely due to attenuation, but it has also been suggested to be a physiologic finding relating to vagal tone [29]), apex, and septum [14,29]. It has also been reported that myocardial MIBG uptake decreases with age in adults- particularly in the later decades of life [26]. Reduced myocardial uptake of MIBG is seen in association with most diseases that result in cardiac injury [31].
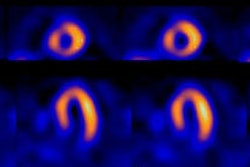
Image interpretation consists of assessment of global tracer uptake on planar images, tracer washout between early and delayed planar images, and regional uptake on tomographic images [33]. Cardiac MIBG uptake is semiquantified by calculating a heart-to-mediastinum ratio on the planar images [15]. The anterior projection seems to be the preferred projection for quantification as it provides the lowest variation and highest resolution [19]. Various methods have been described. In one method a 7 x 7 pixel ROI is placed over the cardiac region and another 7 x 7 pixel ROI over the upper mediastinal area [13]. In another method, left ventricular activity is measured by manual drawn regions of interest surrounding the entire myocardium (excluding ventricular blood pool) [15]. A separate 20 x 20 pixel region of interest is placed over the upper mediastinum. The heart-mediastinum ratio is calculated without background subtraction as mean (or average) counts per pixel over the entire left ventricle divided by mean (or average) counts per pixel in the upper mediastinum [9,15,29]. A heart-to-mediastinum ratio of greater than 1.8 is considered normal [9]. Other authors report a normal HMR as 2.2 +/- 0.3, and a ration of less than 1.6 as abnormal [33].
Another measurement that is calculated is the washout rate [29]. The washout rate (WR), compensated for tracer decay, is thought to reflect turnover of catecholamines and thus sympathetic drive (a measure of the ability of the myocardium to retain MIBG) [29,33]. The clearance rate from the myocardium (washout rate) is calculated by: (Initial myocardial MIBG uptake - Delayed myocardial MIBG uptake/ Initial MIBG uptake) x 100 [13]. The washout rate between early and delayed images should be less than 10% [9]. Non-uniform soft tissue attenuation over the chest can cause variations in the measurements obtained [9].
In healthy subjects, there is low within subject variability in I-123 MIBG uptake on both planar (about 5%) and SPECT (about 5%) imaging [19]. Assuming this is also true for patients with cardiac disease, the effects of therapeutic interventions can be monitored using I-123 MIBG imaging [19].
In cardiomyopathies/heart failure:
Cardiac MIBG uptake is decreased in patients with congestive heart failure, cardiomyopathies, and ventricular arrhythmias [26].
In patients with heart failure the assessment of sympathetic activity has important prognostic implications regarding patient survival and will result in better therapy and outcome [10,32]. Alterations in the cardiac sympathetic nervous system clearly play a role in the development and progression of congestive heart failure (CHF) [13,15]. Altered autonomic function is also responsible for ventricular arrhythmias or sudden cardiac death [25]. Decreased cardiac output related to left ventricular dysfunction results in a baro-receptor mediated increased in sympathetic tone and circulating norepinephrine levels in an effort to compensate for the decreased cardiac output [15,33]. The consequences of chronically increased sympathetic tone include increased left ventricular afterload, worsening LV function, and progressive CHF [15]. The hyperactivity of the sympathetic nervous system in patients with chronic CHF leads to a downregulation in myocardial beta-adrenergic receptors [29]. It has been shown that in CHF patients, the postsynaptic beta1-adrenoreceptor density is reduced and the concentration of inhibiting Gi-alpha proteins is elevated [13]. Cardiac noradrenaline turnover is also increased [13]. Beta-adrenergic blocking agents can aid in blocking sympathetic over-stimulation associated with CHF and I-123 MIBG imaging can be used to evaluate the effectiveness of the treatment [15,32].
Cardiac MIBG uptake is generally diffusely decreased in patients with dilated cardiomyopathies (defects in patients with ischemic cardiomyopathies tend to be more severe in necrotic segments) [11,14]. In these patients, low MIBG uptake is associated with an increased risk for cardiac death [9,11,14,15]. Decreased MIBG uptake on delayed images is closely related to the degree of LV dysfunction in patients with heart failure and also correlates with the level of decreased exercise capacity [8,16]. The poor tracer uptake is likely reflective of underlying cardiac autonomic dysfunction (areas of denervation hypersensitivity) which may place patients at higher risk for arrhythmogenesis [11,35]. The delayed H/M ratio has been reported to be the best predictor for survival in patients with CHF and reduced cardiac function [23]. In patients with CHF, a normal HMR predicts a <1% yearly risk of cardiac death, while a decreased HMR predicts a poor prognosis [33]. In one study of CHF patients, a HMR<1.2 was associated with a 12 month survival of only 40% [33]. Improvement in MIBG uptake following initiation of beta-blocker therapy may predict which patients are more likely to respond to this form of therapy- even prior to documented LV function improvement [14]. Patients that fail to demonstrate an improved HMR following 6 months of optimal medical therapy are at an increased risk for cardiac death [33].
Tracer washout rates have also been studied. Patients with dilated cardiomyopathies typically have accelerated washout rates of MIBG of greater than 25% between early and delayed images (normal less than 10%) [9]. A washout rate of greater than 50% is also associated with an increased risk for cardiac death [9,11] (other quote greater than 27% in chronic CHF patients being associated with a significant increased risk for sudden death (35% cardiac death rate) [29]). The percent change in washout rate between serial exams may also provide information regarding increased risk for cardiac or sudden death (greater than or equal to -5% is associated with an increased risk) [28]. Increased washout can also be seen in other conditions including myocardial hypertrophy and diabetic hearts [11]. MIBG uptake ratios may also help to predict a good response to alpha-blocker therapy in patients with dilated cardiomyopathies [9].
Cardiac resynchronization therapy (CRT) has been shown to be beneficial in patients with advanced chronic heart failure (LVEF <35%) and a QRS duration greater than 120 milliseconds [22,24]. CRT can improve clinical manifestations and quality of life, reduce hospitalizations for CHF, reduce complications, and risk of death (increase survival) [22,24]. Unfortunately, between 20-30% of patients do not respond to CRT [24]. Lower MIBG uptake (H/M ratio below 1.36) is associated with a higher likelihood for lack of response to CRT [22]. The lower MIBG uptake may reflect hearts with more severe myocardial damage that are less likely to respond to CRT [22]. Following successful CRT intervention, there is improved cardiac uptake of MIBG [22]. The presence of left ventricular dyssynchrony also suggests patients that are more likely to respond to CRT [24]. Phase image analysis from gated cardiac examinations can be used to evaluate for the presence of LV dyssynchrony [24]. Extensive LV scarring - particularly when present in the region in which the LV pacing lead is positioned (typically the posterolateral region)- can also result in a decrease likelihood for response to CRT (extensive scarring is predictive of lack of CRT response with a sensitivity of 83% and a specificity of 74%) [24]. This suggests that improvement in LV function is prohibited in the presence of extensive scar tissue [24].
Patients with hypertrophic cardiomyopathies (HCM) also demonstrate derangements in sympathetic activity [12]. HCM is an inherited cardiac muscle disease that is related to a mutation in the genes that encode proteins in the sarcomere [12]. Charateristics of the disorder include disproportionate left ventricular hypertrophy and diastolic dysfunction [12]. Delayed MIBG images demonstrate poor, heterogeneous cardiac tracer retention and increased washout rates [12,29]. In patients with HCM, MIBG washout is significantly higher in patients with ventricular tachycardia (VT) compared to those without VT [29].
In heart transplantation:
The surgical procedure of heart transplantation causes autonomic denervation of the donor (allograft) heart [7]. Immediately after heart transplantation no activity is detected in the myocardium [14]. Total denervation persists for at least 12 months after transplantation [14]. Regional reappearance of sympathetic nerve fibers occurs in the transplanted heart over time [7]. MIBG uptake indicating partial sympathetic reinnervation can be shown in 40% of patients 3 to 5 years after transplantation [7]. Serial MIBG studies show that reinnervation begins from the anterolateral base of the heart and spreads towards the apex [9,14]. With reinnervation, patchy MIBG uptake is seen primarily in the anterior, anterolateral, and septal regions. MIBG uptake is usually not apparent in the posterior or inferior myocardial regions, except for basal posterior localization [9]. Complete reinnnervation of the heart is not seen- even up to 12 years after transplantation [9].
In myocardial infarction/ischemia:
Sympathetic nerve fibers are more susceptible to oxygen deprivation than myocytes and take longer to recover [29,33]. Uptake of I-123 MIBG is decreased in areas of acute and chronic ischemia [9]. Regional denervation of the heart in the post-ischemic myocardium may persist for 15 days or longer following an ischemic event [27]. Sympathetic denervation has also been shown in patients with stable angina in the absence of myocardial infarction presenting as MIBG defects with preserved perfusion [29].
Myocardial infarction destroys the myocardium and the nervous tissue in the myocardium. However, in the early post-infarction period, the MIBG defect is typically larger than the area of infarcted myocardium (a perfusion-innervation mismatch) [9,14]. This is because infarction can result in destruction of proximal neurons which supplied innervation to distal areas in which myocardial tissue is still viable (perfused and viable, but denervated myocardium) [14,33]. As a result, a defect identified on MIBG scintigraphy following infarction, may be more extensive than the actual area of infarction as demonstrated by thallium. Patient's that demonstrate a perfusion-MIBG mismatch (perfused, but decreased MIBG activity) have electrophysiologic abnormalities that can predispose to lethal arrhythmias [33]. Denervated, but viable myocardium has been shown to be supersensitive to catecholamines and this may explain an increased risk for arrhythmogenicity in certain patients following MI [14,29]. Reinnnervation to these peri-infarct regions can be demonstrated by reappearance of MIBG uptake usually within 14 weeks following the cardiac event [9]. Unfortunately, reinnervation may be incomplete [9].
In patients with ventricular tachyarrhythmias:
Electrophysiologic instability is an important trigger of cardiac arrhythmias and is modulated by autonomic function [34]. Abnormally decreased I-123 MIBG uptake can be seen in patients with ventricular tachyarrhythmias and is a powerful predictor of recurrent arrhythmic events [30,34].
Other settings of abnormal myocardial innervation:
Reduction of MIBG uptake in the heart is considered a specific finding for idiopathic Parkinson's disease without autonomic failure and can be used to differentiate it from other parkinsonian syndromes [29].
A significant decrease in MIBG uptake is seen in the inferior and lateral segments in hypertensive patients with cardiac hypertrophy [29].
Cardiac sympathetic denervation/autonomic dysfunction in diabetic patients can also be evaluated with MIBG [29]. Decreased MIBG uptake in diabetics is associated with an increased mortality rate [29].
PET Adrenergic Imaging:
C11-hydroxyephedrine (HED) has been used for mapping cardiac sympathetic activity [29]. The agent is resistant to metabolism by monoamine oxidase and catecholamine O-methyltransferase and has a high affinity for the uptake 1 mechanism [29]. HED is believed to undergo continuous relaease and reuptake by sympathetic neurons [29]. The distribution of the tracer in the myocardium is normally homogeneous without the decreased activity in the inferior wall noted on SPECT MIBG imaging [29]. [F18] 6-fluorodopamine may also be useful for cardiac imaging and the agent has a longer half-life [29]. The tracer is accumulated mainly via the presynaptic uptake 1 mechanism and then sequestered into sotrage vesicles and beta-hydroxylated to fluoroepinephrine [29].
Conditions which decrease cardiac MIBG uptake
- Pheochromocytomas/Elevated circulating levels of norepinephrine which competes with MIBG for the type I uptake system
- After eating
- Administration of yohimbine (due to increased adrenergic activity)
- Administration of tricyclic antidepressants (imipramine, desipramine)
- Administration of sympathomimetics (pseudoephedrine or phenylpropanoloamine)
- Cardiovascular autonomic neuropathy of diabetes mellitus: Cardiovascular autonomic neuropathy is a serious complication of diabetes and the prevalence can be as high as 20-30% of patients with non-insulin dependent diabetes (NIDDM) [21]. Decreased cardiac MIBG uptake in diabetic patients is associated with an increased mortality [9,21]. Improvement in glycemic control has been shown to result in partial restoration of sympathetic innervation [14].
- Following cardiac transplantation (within 1 year, beyond this time about 50% of patients may demonstrate uptake indicative of re-innervation)
- Following ascending aortic aneurysm surgical repair- most likely the result of mechanical damage to the cardiopulmonary nerves surrounding the aorta [10].
- Following chemotherapy with doxorubicin: Decreased myocardial MIBG uptake can be seen following doxorubicin therapy, with limited morphologic damage [9]. Decreased MIBG uptake follows a dose dependent decline with about 25% of patients demonstrating some decrease in MIBG uptake at cumulative doses of 240-300 mg/m2 [9]. Decreased MIBG uptake precedes deterioration of ejection fraction [9,14]. Evidence of sympathetic damage can be used to select patients at risk of severe functional impairment and who may benefit from cardioprotective agents or changes in the schedule of antineoplastic drugs [9].
- Congestive heart failure secondary to pressure or volume overload
- Dilated cardiomyopathy- washout is also increased in these patients [6]
- Acute Myocarditis [6]
- Myocardial infarction [6]
- Sympathetic nerve destruction by stellate ganglionectomy
- Epicardial phenol application, and Shy-Drager syndrome.
- Parkinson's disease [10,14]. MIBG abnormalities observed in Parkinson's patients may be due to postgangliotic sympathetic dysfunction and has been correlated with severity and length of disease [14].
- LV hypertrophy due to essential hypertension [14]. MIBG abnormality is mainly observed in the inferior and lateral walls and the degree of abnormality correlates with the severity of hypertrophy [14].
Conditions which increase cardiac MIBG uptake
- Administration of clonidine (an alpha-2 antagonist which slows nerve traffic)
- Heart failure secondary to coronary artery disease
REFERENCES:
(1) J Nucl Med 1995; Tsuchimochi S, et al. Age and gender differences in normal myocardial adrenergic neuronal function evaluated by iodine-123-MIBG imaging. 36: 969-974
(2) J Nucl Med 1998; Sakata K, et al. Physiologic fluctuation of the human left ventricle sympathetic nervous system assessed by Iodine-123-MIBG. 39: 1667-1671
(3) Nucl Med Annual 1993; Sisson JC. The adrenergic nervous system of the heart and nuclear medicine. Ed. Freeman LM. Raven Press, NY.: 233-249 (No abstract available)
(4) J Nucl Med 1995; Mantysaari M, et al. Myocardial sympathetic nervous dysfunction detected with iodine-123-MIBG is associated with low heart rate variability after myocardial infarction. 36: 956-61
(5) J Nucl Med 1995; Glowniack JV. Cardiac studies with metaiodobenzylguanidine: a critique of methods and interpretation of results. 36: 2133-37 (No abstract available)
(6) J Nucl Med 1998; Agostini D, et al. Impariment of cardiac neuronal function in acute myocarditis: Iodine-123-MIBG scontography study. 39: 1841-1844
(7) J Nucl Med 2001; Odaka K, et al. Reappearance of cardiac presynaptic nerve terminal s in the transplanted heart: Correlation between PET using 11C-hydroxyephedrine and invasively measured norepineephrine release. 42: 1011-1016
(8) J Nucl Med 2001; Zhao C, et al. Comparison of cardiac sympathetic nervous function with left ventricular function and perfusion in cardiomopathies by 123I-MIBG SPECT and 99mTc-Tetrofosmin electrocardiographically gated SPECT. 42: 1017-1024
(9) J Nucl Med 2001; Carrio I. Cardiac neurotransmission imaging. 42: 1062-1076
(10) J Nucl Med 2001; Momose M, et al. Total and partial cardiac sympathetic denervation after surgical repair of ascending aortic aneurysm. 42: 1346-1350
(11) J Nucl Med 2001; Wakabayashi T, et al. Assessment of underlying etiology and cardiac sympathetic innervation to identify patients at high risk of cardiac death. 42: 1757-1767
(12) J Nucl Med 2002; Shimizu M, et al. Heterogeneity of cardiac sympathetic nerve activity and systolic dysfunction in patients with hypertrophic cardiomyopathy. 43: 15-20
(13) J Nucl Cardiol 2002; Parthenakis FI, et al. Segmental pattern of myocardial sympathetic innervation in idiopathic dilated cardiomyopathy: relationship to regional wall motion and myocardial perfusion abnormalities. 9: 15-22
(14) J Nucl Cardiol 2002; Patel AD, Iskandrian AE. MIBG imaging. 9: 75-94 (No abstract available)
(15) J Nucl Cardiol 2002; Gerson MC, et al. Carvedilol improves left ventricular function in heart failure patients with idiopathic dilated cardiomyopathy and a wide range of sympathetic nervous system function as measured by iodine 123 metaiodobenzylguanidine. 9: 608-615
(16) J Nucl Med 2003; Sato M, et al. Correlation between cardiac norepinephrine overflow during exercise and cardiac 123I-MIBG uptake in patients with chronic heart failure. 44: 1618-1624
(17) J Nucl Med 2003; Inoue Y, et al. Effect of collimator choice on quantitative assessment of cardiac iodine 123 MIBG uptake. 10: 623-32
(18) J Nucl Cardiol 2003; Narula J, Sarkar K. A conceptual paradox of MIBG uptake in heart failure: retention with incontinence. 10: 700-704
(19) J Nucl Cardiol 2004; Somsen GA, et al. Normal values and within-subject variability of cardiac I-123 MIBG scintigraphy in heathy individuals: implications for clinical studies. 11: 126-133
(20) J Nucl Cardiol 2004; Flotats A, Carrio I. Cardiac neurotransmission SPECT imaging. 11: 587-602
(21) J Nucl Cardiol 2006; Mushtaq N, et al. I-123 metaiodobenzylguanidine imaging in non-insulin-dependent diabetic patients with normal myocardial perfusion scans: new insights into their increased cardiac morbidity and mortality rates. 13: 8-10
(22) J Nucl Cardiol 2007; D'Orio Nishioka SA, et al. Cardiac sympathetic activity pre and post resynchronization therapy evaluated by 123I-MIBG myocardial scinitgraphy. 14: 852-859
(23) J Nucl Med 2007; Kasama S, et al. Additive effects of spironolactone and candesartan on cardiac sympathetic nerve activity and left ventricular remodeling in patients with congestive heart failure. 48: 1993-2000
(24) J Nucl Med 2007; Henneman MM, et al. Nuclear imaging in cardiac resynchronization therapy. 48: 2001-2010
(25) J Nulc Med 2008; Nagahara D, et al. Predicting the need for an implantable cardioverter defibrillator using cardiac metaiodobenzylguanidine activity together with plasma natriretic peptide concentration or left ventricular function. 49: 225-233
(26) J Nucl Cardiol 2008; Chen W, et al. Age-related decrease in cardiopulmonary adrenergic neuronal function in children as assessed by I-123 metaiodobenzylguanidine imaging. 15: 73-79
(27) J Nucl Med 2008; Vesely MR, Dilsizian V. Nuclear cardiac stress testing in the era of molecular medicine. 49: 399-413
(28) J Nucl Med 2008; Kasama S, et al. Prognostic value of serial cardiac 123I-MIBG imaging in patients with stabilized chronic heart failure and reduced left ventricular ejection fraction. 49: 907-914
(29) J Nucl Cardiol 2008; Henneman MM, et al. Cardiac neuronal imaging: application in the evaluation of cardiac disease. 15: 442-55
(30) J Nucl Med 2009; Akutsu Y, et al. The significance of cardiac sympathetic nervous system abnormality in the long-term prognosis of patients with a history of ventricular tachyarrhythmia. 50: 61-67
(31) J Nucl Cardiol 2009; Jacobson AF, et al. 123I-mIBG scintigraphy to predict risk for adverse cardiac outcomes in heart failure patients: design of two prospective multicenter international trials. 16: 113-121
(32) J Nucl Cardiol 2009; Flotats A, Carrio I. Radionuclide noninvasive evaluation of heart failure beyond left ventricular function assessment. 16: 304-315
(33) J Nucl Cardiol 2010; Yong S, Travin MI. Radionuclide imaging of cardiac autonomic innervation. 17: 655-666
(34) J Nucl Med 2010; Nishisato K, et al. Impaired cardiac sympathetic innervation and myocardial perfusion are related to lethal arrhythmia: quantification of cariac tracers in patients with ICDs. 51: 1241-1249
(35) J Nucl Cardiol 2011; Tamaki N, et al. Novel iodinated tracers, MIBG and BMIPP for nuclear cardiology. 18: 135-143