A research team in Finland has validated the accuracy of a SPECT/CT measure for diagnosing transthyretin amyloidosis (ATTR).
The measure is called the “myocardium-to-blood ratio” (MBR) and was 100% accurate for detecting ATTR cardiomyopathy in a group of patients with suspected disease, noted a group led by Olli Suomalainen, MD, of the University of Helsinki.
“The MBR measurement can be obtained in any SPECT or SPECT/CT system. Thus, our results could be adapted into clinical practice at any nuclear imaging department,” the group wrote. The study was published March 28 in the Journal of Nuclear Cardiology.
ATTR is a progressive condition in which abnormal protein deposits called amyloids build up in the body's organs and tissues, including the heart. Currently, the diagnosis of ATTR cardiomyopathy on nuclear imaging is based on semiquantitative assessments, with quantitative SPECT imaging yet to be translated into clinical practice, the authors explained.
Thus, in this study, they aimed to provide a simple and optimal quantitative SPECT/CT measurement for diagnosing the disease using technetium-99m hydroxymethylene diphosphonate (Tc-99m HMDP) radiotracer. The MBR is calculated by comparing tracer uptake in the heart to that in the blood.
The study included 80 patients (mean age, 80) who underwent Tc-99m HMDP SPECT/CT for suspected ATTR cardiomyopathy between 2018 and 2023, 32 of whom were diagnosed with the disease. The researchers calculated the MBR in the left ventricle (LV) using maximum standardized uptake values for the radiotracer.
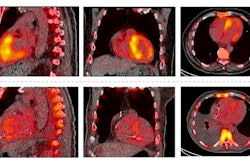
 Typical transthyretin amyloidosis cardiomyopathy (ATTR), early detected ATTR, and a non-ATTR on cardiac MRI and SPECT/CT. Four-chamber cardiac MRI cine image (A), typical diffuse late gadolinium enhancement on cardiac MRI (B), and positive SPECT/CT (C) in a typical ATTR cardiomyopathy patient. The white outline denotes the whole left ventricular (LV) volume of interest (VOI) to obtain maximum myocardial standardized uptake value (SUVmax). Black and yellow circles demonstrate left and right atrial blood pool regions of interest. Four-chamber cardiac MRI cine image (D), nontypical septal predominant late gadolinium enhancement on cardiac MRI (E), and septal SPECT/CT uptake in a patient with early detected ATTR (white asterisk). The red arrow points to a minor right ventricular signal. Negative control patient on cardiac MRI (G and H) and SPECT/CT (I). The white line denotes the LV region of interest in SPECT/CT-negative patient. ECV = extracellular volume; LV MBR = left ventricular myocardium-to-blood ratio. Image and caption available for republishing under Creative Commons license (CC BY 4.0 DEED, Attribution 4.0 International) and courtesy of the Journal of Nuclear Cardiology.
Typical transthyretin amyloidosis cardiomyopathy (ATTR), early detected ATTR, and a non-ATTR on cardiac MRI and SPECT/CT. Four-chamber cardiac MRI cine image (A), typical diffuse late gadolinium enhancement on cardiac MRI (B), and positive SPECT/CT (C) in a typical ATTR cardiomyopathy patient. The white outline denotes the whole left ventricular (LV) volume of interest (VOI) to obtain maximum myocardial standardized uptake value (SUVmax). Black and yellow circles demonstrate left and right atrial blood pool regions of interest. Four-chamber cardiac MRI cine image (D), nontypical septal predominant late gadolinium enhancement on cardiac MRI (E), and septal SPECT/CT uptake in a patient with early detected ATTR (white asterisk). The red arrow points to a minor right ventricular signal. Negative control patient on cardiac MRI (G and H) and SPECT/CT (I). The white line denotes the LV region of interest in SPECT/CT-negative patient. ECV = extracellular volume; LV MBR = left ventricular myocardium-to-blood ratio. Image and caption available for republishing under Creative Commons license (CC BY 4.0 DEED, Attribution 4.0 International) and courtesy of the Journal of Nuclear Cardiology.
According to the analysis, the LV MBR was higher in ATTR patients than non-ATTR patients (4.6 vs. 0.9, p < 0.001). Further, the group determined that an LV MBR greater than 1.5 was the optimal cut-off value to differentiate ATTR from non-ATTR patients with an accuracy of 100%.
“We believe that adapting SPECT/CT-derived diagnostic criteria for ATTR diagnosis would increase the sensitivity of amyloid imaging and provide a simple approach to study interpretation, which would streamline the current diagnostic pathway,” the group wrote.
Ultimately, there are two types of ATTR, a hereditary type caused by a genetic mutation and a “wild-type” typically associated with aging. The patients in this study were wild-type ATTR patients, as the hereditary form is rare in Finland, which was a limitation of the study, the researchers noted.
Nonetheless, the study showed that quantitative Tc-99m HMDP SPECT/CT using MBR is a feasible and accurate method for diagnosing ATTR cardiomyopathy and warrants further research in prospective trials, they wrote.
“Further work is still needed to translate universally accepted SPECT/CT measures to daily nuclear medicine practice,” the group concluded.
The full study is available here.












![A 53-year-old patient (patient number four) with a recurrent pituitary adenoma with extension of a cystic component of disease to the medial temporal lobe apparent on MRI (contoured in blue), and extension of disease to the left sphenoid bone and orbital apex apparent on [68Ga]Ga-DOTA-TATE (contoured in yellow).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/04/pituitary-tumor.QGsEnyB4bU.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)