A new analytic framework for brain PET has produced an AI biomarker that when applied to Alzheimer's disease better aligns imaging findings with neurodegenerative and clinical outcomes, according to a study published April 7 in Radiology.
Called interpretable adversarial decomposition learning (ADL), the method addresses a gap where "biologic positivity" translates (or fails to translate) into Alzheimer's disease neurodegenerative and clinical expression, according to lead authors Cheng Tang, Xun Sun, MD, PhD, and colleagues at Huazhong University of Science and Technology in Wuhan, China.
"Our work is positioned within a broader shift toward quantitative PET measures that are sufficiently standardized and interpretable for routine clinical use," stated the authors.
While template-based PET metrics quantify Alzheimer's disease (AD) amyloid-β (Aβ) and tau burden, they compress whole-brain data into a single scalar, overlooking disease heterogeneity and sometimes causing imaging–clinical discordance, according to the group. In their new approach, the researchers developed and validated ADL for separating AD-specific pathologic signal from physiologic uptake at the individual level.
They used data obtained from the U.S. site of the Alzheimer’s Disease Neuroimaging Initiative (ADNI), the Australian Imaging, Biomarkers and Lifestyle (AIBL) study, the Global Alzheimer’s Association Interactive Network databases, and Wuhan Union Hospital in Shanghai.
External testing of Aβ models included information from a subset of patients from the Global Alzheimer’s Association Interactive Network Centiloid Project. Tau PET models were developed analogously using ADNI tau and local datasets, with external testing on CenTauR Project data.
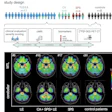
The retrospective study analyzed 7,457 amyloid-β [Aβ] PET scans from 3,595 patients and 1,894 tau PET scans from 1,127 patients to assess ADL's clinical relevance relative to conventional metrics.
According to the findings, the AI model achieved strong diagnostic accuracy, with area-under-the-curve (AUC) values of 0.94 for amyloid and 0.98 for tau. External testing AUCs were 0.94 for Aβ and 0.98 for tau.
ADL also generated voxel-level pathologic maps and Alzheimer disease adversarial decomposition (ADAD) scores that demonstrated independent baseline and longitudinal associations with cognitive outcomes and hippocampal atrophy after adjustment, according to the group.
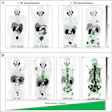
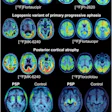
 Adversarial decomposition learning (ADL) enables intuitive, longitudinal visualization of disease progression and treatment response, supporting clinician–artificial intelligence collaboration. (A) Images in a 48-year-old female patient who presented with mild cognitive impairment. The ADL probability maps highlighted occipital-temporal amyloid β (Aβ) spread and focal left-temporal tau pathologic abnormalities. An area of increased occipital AV45 uptake is shown (arrowhead). (B) Images in a 51-year-old male who presented with memory decline. For each image grid, the left side is the decoupler-output AD pathologic probability map, the upper right side is the original Aβ/tau PET image, and the lower right side is the image after AD component removal by the decoupler.Image and caption courtesy of RSNA.
Adversarial decomposition learning (ADL) enables intuitive, longitudinal visualization of disease progression and treatment response, supporting clinician–artificial intelligence collaboration. (A) Images in a 48-year-old female patient who presented with mild cognitive impairment. The ADL probability maps highlighted occipital-temporal amyloid β (Aβ) spread and focal left-temporal tau pathologic abnormalities. An area of increased occipital AV45 uptake is shown (arrowhead). (B) Images in a 51-year-old male who presented with memory decline. For each image grid, the left side is the decoupler-output AD pathologic probability map, the upper right side is the original Aβ/tau PET image, and the lower right side is the image after AD component removal by the decoupler.Image and caption courtesy of RSNA.
Centiloid (CL) and CenTauRz (CTRz) can provide robust population-level anchors for biologic characterization, whereas ADAD may be more sensitive to clinically meaningful expression of disease in individual patients, they noted. A key finding from the study was that ADAD showed larger effect sizes with cognition and hippocampal volume than established scales in mixed-effects modeling, suggesting closer coupling to functional status and neurodegeneration, the authors wrote.
While established quantitative scales correlated more strongly with autopsy findings and cerebrospinal fluid (CSF) biomarkers, the AI-derived scoring tool still showed independent associations with cognitive decline and hippocampal volume loss over time, the group added.
The researchers said ADL and voxel-wise attribution maps may complement visual reads and facilitate shared interpretation among nuclear medicine physicians, neurologists, and patients. The framework may be attractive for settings where chemical solutions to off-target binding are difficult to implement in practice, enabling post-hoc mitigation at the image level, they added.
Future work, however, should explore biomarker-centric or autopsy-informed reference definitions, where available, according to the group, which also suggested that future prospective studies and broader external testing (including with additional tau tracers) are needed to characterize generalizability.
Read the complete paper here.