A multicenter study has found that the incidence of nephrogenic systemic fibrosis (NSF) was much greater at two centers that used gadodiamide, compared to two facilities that used gadopentetate dimeglumine.
Researchers from the University of North Carolina in Chapel Hill, Emory University in Atlanta, Thomas Jefferson University in Philadelphia, and Wake Forest University in Winston-Salem, NC, collaborated on the study, which was published online in Radiology (July 15, 2008). NSF is a debilitating skin condition that has developed in a number of patients with renal impairment who received gadolinium-based contrast.
The retrospective study reviewed patients who were diagnosed with NSF between January 2000 and December 2006 to determine the benchmark incidence of NSF based on the use of different gadolinium chelate contrast agents at the four tertiary-care centers. Researchers developed a standard checklist for each facility to obtain data on the use and administration of gadolinium chelate contrast agents and the development of NSF.
The study also assessed serum creatinine level and functional renal status of each patient through laboratory and medical records at or nearest the time of gadolinium chelate contrast agent administration prior to the onset of NSF.
Agents in use
During the seven years under review by the researchers, the University of North Carolina and Emory University used gadodiamide (Omniscan, GE Healthcare, Chalfont St. Giles, U.K.), while Wake Forest University and Thomas Jefferson University used gadopentetate dimeglumine (Magnevist, Bayer HealthCare Pharmaceuticals, Wayne, NJ).
Of 28 patients who had a diagnosis of NSF at the University of North Carolina, 23 patients had undergone gadolinium chelate-enhanced MRI. Two of the remaining five patients received gadolinium chelate-enhanced MRI at another facility before developing NSF, but the type of gadolinium chelate contrast agent that was administered could not be verified. The relationship between NSF and gadolinium chelate contrast agent administration could not be identified in the other three patients.
At Emory University, 14 patients were diagnosed with NSF, with nine of the 14 patients having undergone gadolinium chelate-enhanced MRI. The study could not determine a relationship between NSF and gadolinium chelate contrast agent in the other five cases.
NSF diagnoses
At Wake Forest University, three of six patients diagnosed with NSF had undergone gadolinium chelate-enhanced MR imaging. Two of the remaining three patients had cases related to gadolinium chelate contrast agent administration, having undergone gadodiamide-enhanced MR imaging outside of Wake Forest before developing NSF. The administered dose of gadodiamide could not be determined. The relationship between NSF and gadolinium chelate contrast agent administration could not be determined in the sixth patient.
At Thomas Jefferson University, two patients had a diagnosis of NSF, with one of the two patients having undergone gadolinium chelate-enhanced MRI there. The relationship between NSF and gadolinium chelate contrast agent administration was unknown in the other patient.
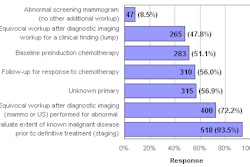
A total of 50 patients among the four institutions were diagnosed with NSF. Thirty-six of those patients (72%) received gadolinium chelate-enhanced MR imaging and were included in the final study population. The patients ranged from 17 to 75 years old, with a mean age of 47.1 years. There were 20 males and 16 females.
Incidence of NSF
The study found that the incidence of NSF varied by institution, based on the type of gadolinium agent used. The benchmark incidence of NSF was one in 2,913 patients at the facilities that used gadodiamide, compared with one in 44,224 patients at facilities using gadopentetate dimeglumine.
At the University of North Carolina and Emory University, where gadodiamide was used, the overall incidence of NSF was 0.039% (32 of 82,260 patients). There was no significant difference in incidence of NSF between the two facilities.
Meanwhile, the overall incidence of NSF at Wake Forest and Thomas Jefferson Universities, which used gadopentetate dimeglumine, was 0.003% (four of 135,347 patients). Again, there was no significant difference in incidence of NSF between the two institutions.
Researchers wrote that the study "confirms the association between the development of NSF and the prior administration of a gadolinium chelate contrast agent in patients with renal insufficiency. [The results] also show that different gadolinium chelate contrast agents are associated with varying benchmark incidences of NSF," adding that the "benchmark incidences of NSF were significantly and remarkably lower at the two centers where gadopentetate dimeglumine was used compared with those at the two centers where gadodiamide was used."
The study also noted that its findings are consistent with previously published research (Radiology, April 2007, Vol. 243:1, pp. 148-157; Nephrology Dialysis Transplantation, February 2004, Vol. 19:2, pp. 469-473; and Journal of Magnetic Resonance Imaging, November 2007, Vol. 26:5, pp. 1198-1203).
Renal compromise
The study also reported that 28 of 36 patients who developed NSF had undergone multiple gadolinium chelate-enhanced MR examinations. That finding, researchers noted, "supports the suggestion that accumulated gadolinium chelate contrast agent, or a cumulative dose, is an important etiologic factor in the development of NSF."
Because 13 of those 36 patients were outpatients, the finding "suggests that NSF may also be seen in patients who have renal compromise with a relatively stable medical condition, in which acute concurrent events may be absent or undetected."
Researchers noted that the study was limited by their inability to track 14 patients (28%) who had NSF but did not undergo MRI. The lack of a centralized medical data registry and patient privacy rights under the Health Insurance Portability and Accountability Act (HIPAA) were two obstacles cited to more complete data collection.
Another limitation was the inability to determine the incidence of NSF in patients who were at risk for NSF, particularly patients with stage 4 or 5 chronic renal disease.
By Wayne Forrest
AuntMinnie.com staff writer
July 24, 2008
Related Reading
Study questions some NSF risk factors, April 15, 2008
Gadolinium/NSF lawsuits centralized in Ohio federal court, March 21, 2008
Contrast firms hit with another NSF lawsuit, March 18, 2008
ECR NSF studies raise as many questions as answers, March 7, 2008
Study offers more evidence that multiple factors up NSF risk, January 24, 2008
Copyright © 2008 AuntMinnie.com