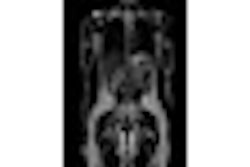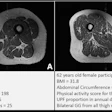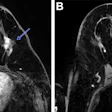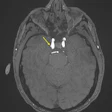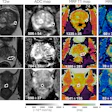
The system features a 70 mtesla/m gradient strength at 200 tesla/m/sec slew rate for enhanced cartilage visualization. The system has received clearance from the U.S. Food and Drug Administration and will be available in the U.S. in December. ONI expects to have worldwide distribution in place by February 2009.