Computer-aided detection (CAD) software based on deep learning trained with MRI data can identify patients with mild cognitive impairment (MCI) due to Alzheimer's disease, according to a study published August 19 in IEEE/ACM Transactions on Computational Biology and Bioinformatics.
The results suggest a noninvasive alternative for identifying and tracking these patients, who are at higher risk of developing Alzheimer's, wrote researchers Füsun Er, PhD, and Dionysis Goularas, PhD, both of Yeditepe University, in Istanbul, Turkey.
"The CAD system yielded a 87.2% accuracy, thus contributing to the efforts toward the creation of a prognosis prediction system which will decide about the type of treatment of MCI patients without necessarily involving the use of invasive methods or cognitive tests," the pair wrote.
Memory decline
Mild cognitive impairment is characterized by a decline in memory, and patients who have MCI often remain stable for years, the researchers noted. But the condition has been linked to increased risk of developing Alzheimer's.
"Although MCI and Alzheimer's disease are distinct disorders, because of the relation between them, it has been of great interest to predict the risk of developing Alzheimer's disease for MCI patients," Er and Goularas wrote.
In addition to neuroimaging such as PET, identifying MCI due to Alzheimer's disease is often done using techniques such as cerebrospinal fluid (CSF) protein or blood tests. But noninvasive methods are better, particularly in this patient population.
"Although several modalities are being used to develop biomarkers for dementias, it should be considered preferable to use noninvasive methods because ethical issues may arise when considering the use of them in the diagnosis and treatment of a patient with impaired mental function," the investigators wrote.
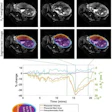
MRI can show changes in gray and white matter tissue, particularly hippocampal and entorhinal atrophy, that point to risk of patients with MCI developing Alzheimer's disease. But identifying these changes -- which can be subtle -- may prove challenging via clinicians' visual assessment alone. So Er and Goularas developed a computer-aided diagnosis system using deep learning to identify MCI due to Alzheimer's disease using data taken from MRI brain exams.
The study included 294 patients with MCI selected from the Alzheimer's Disease Neuroimaging Initiative (ADNI), a study launched in 2003 to investigate clinical, imaging, genetic, and biochemical biomarkers for the early detection and tracking of Alzheimer's disease. Of the 294 patients included in the study, 125 developed Alzheimer's disease during follow-up, while the rest remained stable
The CAD model used MRI data from two periods: baseline and 12-month follow-up. It took the "differences between the two MRI volumes and [generated] a new one ... detecting the regions of interest that are statistically important between the MCI patients who remained stable and those who developed Alzheimer's disease," Er and Goularas wrote.
The authors found that the MRI-trained CAD software distinguished patients with MCI who developed Alzheimer's disease from those who did not with a sensitivity rate of 92.4%, an accuracy rate of 87.2%, and a specificity rate of 80.4%.
The CAD system did not necessarily outperform more invasive techniques for identifying MCI, but it was comparable enough to other techniques as to be a solid alternative.
"In comparison with other systems, [our] results were higher from all systems that use only MRI data, lower only than those that utilized invasive methods like CSF tests [and] minimally invasive methods like plasma, and ... similar to studies that used additional cognitive tests," the authors wrote. "[Our] proposed method presents a significant advantage as it does not need invasive procedures which may be uncomfortable, inappropriate, or ... difficult to be executed [in an elderly population]."
Better treatment
Better prediction of Alzheimer's disease risk in patients with MCI could translate to more effective treatment, according to the investigators.
"Using a system for the prognostic diagnosis of Alzheimer's disease in MCI patients is vital to start the treatment immediately using preventive or therapeutic medicines for slowing down the progression to Alzheimer's disease or treating the symptoms of dementia," Er and Goularas concluded. "[However] the use of preventive medication in MCI patients who actually will not develop Alzheimer's disease may expose those patients to the unnecessary burden of medication. For this reason, identifying MCI patients who are most likely to convert to Alzheimer's disease with a high accuracy is essential."














![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)