Lobular carcinoma in situ (LCIS) is both a cancer precursor indicator and a marker for increased breast cancer risk, according to findings presented May 4 at the Society of Breast Imaging (SBI) annual meeting in National Harbor.
Dr. Gary Whitman from the University of Texas MD Anderson Cancer Center shared findings from a study that showed that the presence of LCIS indicates a substantial immediate risk of breast cancer at the site of it as well as a sustained and persistent risk of cancer in both breasts.
"Our findings show that pathologists' determination of non-classic [LCIS] variants [florid and pleomorphic with higher grades and/or necrosis] should undergo excision," co-author Dr. Hannah Chung, also of MD Anderson, told AuntMinnie.com. "Upgrade to cancer and eventual development of cancer is highest at the site of LCIS, in the same breast [as opposed to the] contralateral breast."
Although rare, the presence of LCIS on surveillance imaging can indicate increased risk of a woman developing invasive breast cancer. In this situation, management strategies include excision, no excision but rather surveillance, or something in between. However, Chung said these all come with a degree of controversy.
Whitman's team sought to determine LCIS outcomes according to the initial recommendation for surgical excision versus imaging surveillance via a study that included 116 LCIS lesions.
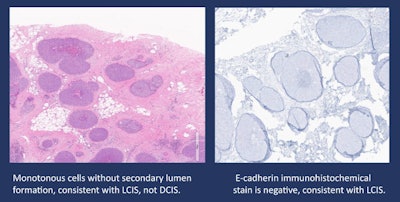 Pleomorphic lobular carcinoma in situ and ductal carcinoma in situ may be confused from each other as was the original pathology interpretation in this case of a 50-year-old woman presenting with suspicious imaging findings on DBT and subsequent ultrasound. Researchers from the University of Texas MD Anderson Cancer Center said these can be differentiated by the use of immunohistochemical stain which shows lack of E-cadherin. Image and caption courtesy of Dr. Hannah Chung.
Pleomorphic lobular carcinoma in situ and ductal carcinoma in situ may be confused from each other as was the original pathology interpretation in this case of a 50-year-old woman presenting with suspicious imaging findings on DBT and subsequent ultrasound. Researchers from the University of Texas MD Anderson Cancer Center said these can be differentiated by the use of immunohistochemical stain which shows lack of E-cadherin. Image and caption courtesy of Dr. Hannah Chung.Of these lesions (which were targeted for percutaneous needle biopsy) 58 had calcifications (50%), 30 showed enhancement on MR imaging (25.9%), and 28 were noncalcified mammographic architectural distortions or masses (24.1%). After multidisciplinary review, surgical excision was recommended for 48 of the lesions (41.4%) and imaging surveillance in the remaining 68 lesions (58.6%).
Of the 116 lesions, 22 were malignant; 11 of these occurred among those recommended for immediate surgical excision at the site of LCIS and another 11 occurred among those recommended for further surveillance. Of these cancers, 14 (63.6%) developed at the site of LCIS, while the other eight developed at a location away from the LCIS site.
The researchers also found that among cases determined to require surgical excision, there was a 22.9% (11/48) upgrade rate. Additionally, if the criteria for surgical excision were not met, the delayed risk of malignancy was 16.2% (11/68) and persisted for up to 13 years.
The team suggested that based on these results, LCIS cases that are not excised can be tracked via imaging surveillance and treated with tamoxifen or an aromatase inhibitor.
These results point to multidisciplinary review playing a critical role in triaging patients diagnosed with LCIS, Whitman noted. He said takes the adequacy of sampling relative to the extent of disease noted on both imaging and pathology and the degree of necrosis and pleomorphism into consideration.
Chung told AuntMinnie.com that while the MD Anderson Cancer Center has a multidisciplinary team to review such cases, radiologists who work with less resources or team participation may be able to determine LCIS cases by applying the following three parameters.
"They can assess how adequate the lesion of interest was, whether the lesion is concordant or discordant, and how significant the atypia is on pathology," she noted.




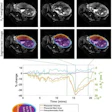
![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=100&q=70&w=100)







![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)