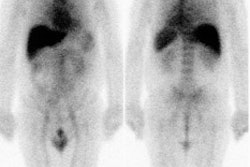
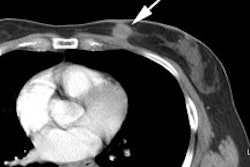
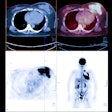
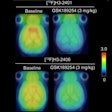
Musculoskeletal Tumors:
Sarcomas are a complex group of tumors [24]. Women are affected
more than men (60%) and most adult soft-tissue sarcomas (60%)
occur in an extremity [24]. Histologic grade reflects biologic
behavior of bone and soft-tissue sarcomas and is incorporated into
their staging classification [14]. Prognostic factors affecting
survival of patients with soft-tissue sarcomas are a deep tumor
location, a tumor 5 cm or larger in greatest dimension, a tumor
located proximally in a lower extremity, and microscopic or
grossly involved tumor margins at resection [24]. In most
patients, once metastases occur, survival diminishes rapidly [24].
FDG uptake is typically related to degree of tumor dedifferentiation and aggressiveness and FDG-PET can be used to evaluate musculoskeletal tumors [18]. FDG exams can detect distant metastases, evaluate for recurrence, and monitor response to therapy. PET/CT has been shown to be more accurate than PET alone for the staging of bone and soft tissue scaromas [14]. Combined PET/CT imaging resulted in correct N-staging in 96% and correct M-staging in 91% of patients, compared to 91% and 79% respectively for conventional imaging alone [14]. Combine PET/CT and conventional imaging results in overall correct staging in up to 87% of patients [14]. Combined PET/CT and conventional imaging can help to alter treatment in up to 14% of patients (compared to conventional imaging alone) and preclude surgical resection in up to 13% of patients [14].
Primary tumor evaluation:
SUV values can be helpful in distinguishing benign from malignant lesions. However, increased FDG uptake in a skeletal lesion is not pathognomonic for a malignant neoplasms [5,8]. Increased tracer accumulation has been described with benign bone lesions (including non-ossifying fibroma, fibrous dysplasia, giant cell tumors, eosinophilic granuloma, and aneurysmal bone cyst) and at sites of inflammation or infection. For sarcomas, the level of FDG uptake reflected by the SUV value is indicative of tumor aggressiveness [18]. Hetergeneous uptake within a tumor has also been suggested to be a negative prognostic indicator [18].
PET FDG false negative examinations have been reported in association with malignant lesions- particularly chondrosarcomas and plasmacytomas, but also low grade osteosarcomas and Ewing's sarcoma [1,5]. False-negative exams can also occur for low grade soft-tissue sarcomas which tend to have low FDG uptake [3].
In a meta-analysis of FDG PET data in the evaluation of
soft-tissue lesions the sensitivity and specificity for malignant
versus benign lesions was 87% and 79%, respectively using an SUV
value of 2.0 (visual quantification was 92% and 73% respectively)
[8]. FDG images are generally positive in intermediate
and high grade tumors (CI: 97-100%), but uptake is less
consistent in low grade lesions (74%, CI: 59-86%) [8].
The presence of intratumoral necrosis and the
volume of necrosis (over 30%) identified on FDG PET/CT imaging
are strong adverse prognostic factors for disease recurrence and
death in patients with limb and girdle sarcomas [26].
Response to therapy:
Perhaps, one of the more important applications of FDG PET is in evaluation of response to therapy [17]. Neoadjuvant chemotherapy and radiation can be used pre-operatively to decrease tumor size in order to facilitate the limited resection needed for limb-salvage surgery [2,17]. The correlation between histopathologic tumor necrosis after preoperative chemotherapy with clinical prognosis is well established [2]. Unfortunately, for soft tissue sarcomas the anatomic definition of partial response by demonstration of a 30% decrease in the sum of the largest diameter of the target lesions is often not able to accurately assess response to treatment [17]. In patients with soft tissue sarcomas, changes in CT-based tumor volumes often fail to discriminate between responders and non-responders [17].
Functional imaging with FDG PET allows detection of biochemical changes within the lesion prior to visible morphological changes. Changes in SUV can predict treatment response [17]. A decrease in the tumor to non-tumor uptake ration of greater than 30% after chemotherapy has been shown to correlate well with extensive histologic tumor necrosis [2]. For patients that fail to demonstrate a response to treatment, early therapy modification can be performed. This is important as post-operative modifications in therapy regimens for non-responders have not been shown to improve patient outcome [2].
FDG PET imaging is superior to bone scintigraphy for evaluation of tumor response to therapy as bone healing (sclerosis) will result in increased Tc-99m-accumulation ("flare" phenomenon) [2].
Following completion of therapy, MR imaging can often be equivocal in differentiating post-therapy changes from tumor recurrence. FDG PET imaging has shown promise in the ability to identify recurrent tumor [7]. When positive in these cases, results of the PET exam can be used to guide biopsy of the most metabolically active regions [7].
18F-labeled NaF (sodium fluoride) may hold promise for the identification of bone metastases with improved lesion detection compared to bone scan [4].
Specific tumors:
Osteosarcoma:
Osteosarcoma is the most common primary malignant bone tumor in children and adolescents (peak incidence in the second decade of life) [16,24]. The metaphysis of long bones is the most frequent site- most commonly about the knee (40-45% of cases involve the distal femur) [16]. Initial tumor stage (histologic grade, size of the primary tumor, and the presence of metastases) and tumor necrosis following neoadjuvant therapy are the strongest prognostic factors for survival [19,20]. Up to 15-20% of patients have pulmonary metastases at the time of diagnosis [24]. The 5-year event-free survival in patients with localized disease is up to 70%, but survival decreases to 30% in patients that present with metastatic disease [16]. A 95% or higher degree of tumor necrosis following neoadjuvant therapy is considered a good response, 90-95% necrosis a partial response, and less than 90% necrosis as nonresponder [21]. Suboptimal histologic response to neoadjuvant chemotherapy, defined as less than 90% tumor necrosis, can be an indication for more intensive postoperative regimens [19]. While progression of the primary tumor on chemotherapy can be an indication for amputation rather than limb salvage [19]. Progression of metastases while receiving preoperative chemotherapy is an indication for non-resection- thereby sparing the patient a futile and morbid procedure [19].
Combined Fluorine-18 (blood flow and osteoblastic response) and FDG (metabolism) imaging can be combined to evaluate primary bone neoplasms. In patients with osteosarcoma, increased uptake of both agents is identified in a distribution corresponding to the bone and soft tissue components of the tumor respectively.
In general, there is good correlation between the amount of FDG
uptake and the histologic grade of the lesion. A high tumor FDG
uptake has also been shown to correlate with a poor prognosis [6],
although other authors indicate that the prognostic value of SUV
is not proven for patients with osteosarcoma [28]. SUVpeak, MTV,
and TLG at baseline, interim, or followup scans have been shown to
be predictive of event free and overall survival [29]. An SUVpeak
< 7.98 at baseline has been associated with improved overall
survival [29]. Metabolic tumor volume is defined as the volume of
tumor tissue with an SUV above a minimal threshold and this
reflects the biologically active tumor burden [28].
Metabolic tumor volume (using a fixed standard uptake value
threshold of 2.0) has been shown to predict a poor response to
neoadjuvant chemotherapy and shortened metastasis-free survival
[28]. Other authors indicate that a MTV (SUV 2.5) < 238 at
baseline is associated with improved overall survival [29]. Total
lesion glycolysis is defined as the product of the mean tumor SUV
and the metabolic tumor volume (above a minimum SUV threshold) and
this has also demonstrated prognostic significance for
osteosarcoma [28]. In one study, a TLG (2.5) < 1022 at baseline
was associated with improved overall survival [29].
FDG PET accurately reflects response of osteosarcoma to
chemotherapy (a decrease in cellularity and loss of viable cells
results in decreased FDG uptake) [19,27]. Following successful
neoadjuvant chemotherapy tumor FDG uptake will decrease [6]. One
article suggested a post treatment SUV max less than 3.3 is
associated with tumor necrosis greater than 90% and is a predictor
of better progression-free and overall survival [19]. In a
prospective study, a post-treatment SUVmax of less than 2 (or a
post treatment/pre treatment SUVmax ratio of less than 0.4) was
associated with a good histologic response [20]. A high SUV max
after chemotherapy is associated with poor survival [19]. However,
inflammatory infiltrates or reactive fibrosis may result in tracer
uptake and false positive findings[27].
18F uptake will still be seen in areas corresponding to sclerosis, but regional accumulation of FDG in these areas will be normal [4].
Ewing sarcoma:
Ewing sarcoma is the second most common malignant bone tumor
found in children and adolescents (peak incidence around the age
of 15 years and 95% of cases between the ages of 4 and 25 years)
[15,24, 25]. Overall, ot is the fourth most frequent primary
malignant tumor of bone after multiple myeloma, osteosarcoma, and
chondrosarcoma [25]. The most common sites of involvment are the
ilium/pelvis (21-26% of cases), the femur (20-21%),
tibia-fibula(8-18%), ribs/chest wall (8-16%), upper extremity
(9-10%), and the spine (6%) [22,25]. Long tubular bone involvement
is more common proximally and is often metadiaphyseal [25].
Patients usually present with non-specific symptoms such as pain (82-88%), a mass or swelling (60%), fever (20-49%), and an increased ESR (43%) [25]. The survival rate is about 60-65% for localized disease, but the 5 year survival decreases to 40% in the presence of lung metastases and 20% for patients with bone metastases [15,22]. The presence of absence of metastatic disease and tumor size have generally been considered the two most important criteria affecting prognosis [22]. Patients generally receive multimodality treatment with chemotherapy, surgery, and radiation [15].
On plain film, bone destruction with a moth-eaten to permative
pattern is seen in 76-82% of lesions, but sclerotic compoenents
can be present in 32-40% of cases [25]. Periosteal changes are
frequent (58-84%) and is usually lamellated or spiculated in
appearance [25].
FDG PET imaging has been shown to be significantly more sensitive
for the detection of osseous metastases compared to bone scan
(sensitivity PET 88% versus 37%) [22,25]. PET/CT has been shown to
be more accurate than stand alone PET for the detection and
localization of lesions in patients with Ewing sarcoma [15]. The
CT portion of the examination permits detection of small lung
nodules that may not be identified on the PET exam [15].
Overall, PET/CT results in improved patient staging [15].
Following induction therapy, a decrease in tumor SUV max to below
2-2.5 has been shown to be associated with a favorable response to
therapy (less than 10% viable tumor) and improved progression-free
survival [22]. Other authors suggest that a reduction in metabolic
tumor burden (MTV) is a better predictor of treatment response
than changes in SUV and that a 90% reduction in MTV was predictive
of a good response to chemotherapy [25]. FDG PET also demonstrates
high accuracy in detection of tumor recurrence locally and
distally [25].
Extraskeletal Ewing sarcoma - the rpesence of extraskeletal
Ewings sarcoma is about 15-20% and is usually seen in young
patients with 85% of cases detected between 20 months and 30 years
of age [25]. The lesion is rare in blacks [25]. Patients often
have a large, rapidly growing, soltary, deep soft-tissue mass
[25]. The most common location is the paravertebral region (32%),
lower extremities (26%), chest wall (18%), retroperitoenum (11%),
pelvis (11%), and upper extremities (3%) [25].
Giant cell tumors:
Giant cell tumors are usually classified as benign lesions, however, they frequently demonstrate local recurrence (up to 50%) and metastatses may occur [9]. Giant cell tumors have generally enhanced FDG uptake- mean SUV 4.8 [9].
Childhood sarcomas:
Soft-tissue sarcomas account for 6-10% of all chidlhood
malignancies and rhabdomyosarcoma constitutes approximately 60% of
these [23]. Certain risk factors as associated with prognosis for
rhabdomyosarcoma and include tumor size and invasiveness, primary
site, gross incomplete surgical removal of localized tumor, age at
presentation, alveolar or embryonal histology, and the presence or
absence of metastatic disease [23]. The 5 year survival for
patients with rhabdomyosarcoma and distant metastases is as low as
25% [23]. In patients with rhabdomyosarcoma, findings associated
with a lower survival rate on PET imaging are higher intensity
tracer uptake within the tumor, metabolically active lymph nodes,
and distant metastases [23].
FDG PET imaging can also detect unsuspected sites of metastatic disease and can be used to monitor response to therapy [10].
Benign fibrous lesions:
Benign fibro-osseous lesions such as non-ossifying fibromas or fibroxanthomas (which are referred to as a fibrous cortical defect when smaller than 3 cm in size [13]), fibrous cortical defects, and cortical desmoids can all demonstrate FDG uptake [11,12,13]. Non-ossifying fibromas and fibrous cortical defects occur in 20-40% of children and are most commonly located in the metaphysis or diametaphyseal junction of the distal femur or proximal tibia [11]. The lesions appear as a radiolucent defect with a thin sclerotic border that is usually scalloped and slightly expansile [13]. The lesions generally undergo progressive ossification/spontaneous regression over time and are often no longer apparent after the second decade of life [11,13]. During their involutional phase, there is increased osteoblastic activity as the lesion is replaced by new bone [11]. Non-ossifying fibromas tend to take longer to regress than do fibrous cortical defects [11]. Cortical desmoids are located on the posteromedial surface of the distal femur at the site of attachment of the extensor tendinous fibers of the adductor magnus muscle- they are the result of repetitive traction microavulsions with a subsequent fibroblastic response [11]. Cortical desmoids are found in about 11% of boys and 4% of girls [11].
All of the above fibroosseous lesions can appear metabolically active on FDG PET imaging [11]. Uptake on the PET scan also does NOT necessarily correlate with increased uptake on bone scan (i.e.: the lesion may not appear active on bone scan) [11].
REFERENCES:
(1) J Nucl Med 2000; Schulte M, et al. Grading of tumors and tumorlike lesions of bone: Evaluation by FDG PET. 41: 1695-1701
(2) Clin Nucl Med 2000; Franzius C, et al. Evaluation of chemotherapy response in primary bone tumors with F-18 FDG positron emission tomography compared with histologically assessed tumor necrosis. 25: 874-881
(3) J Nucl Med 2001; Dimitrakopoulou-Strauss A, et al. Dynamic PET 18F-FDG studies in patients with primary and recurrent soft-tissue sarcomas: Impact on diagnosis and correlation with grading. 42: 713-720
(4) J Nucl Med 2001; Schirrmeister H, et al. Prospective evaluation of the clinical value of planar bone scans, SPECT, and 18F-labeled NaF PET in newly diagnosed lung cancer. 42: 1800-1804
(5) J Nucl Med 2002; Dimitrakopoulou-Strauss A, et al. The role of quantitative 18F-FDG PET studies for the differentiation of malignant and benign bone lesions. 178: 510-518
(6) J Nucl Med 2002; Franzius C, et al. Prognostic significance of 18F-FDG and 99mTc-methylene diphosphonate uptake in primary osteosarcoma. 43: 1012-1017
(7) AJR 2002; Bredella MA, et al. Value of FDG positron emission tomography in conjunction with MR imaging for evaluating therapy response in patients with musculoskeletal sarcomas. 179: 1145-1150
(8) J Nucl Med 2003; Ioannidis JPA, Lau J. 18F-FDG PET for the diagnosis and grading of soft-tissue sarcoma: a meta-analysis. 44: 717-724
(9) J Nucl Med 2004; Strauss LG, et al. 18F-FDG kinetics and gene expression in giant cell tumors. 45: 1528-1535
(10) AJR 2005; McCarville MB, et al. PET/CT in the evaluation of childhood sarcomas. 184: 1293-1304
(11) AJR 2006; Goodin GS, et al. PET/CT characterization of fibroosseous defects in children: 18F-FDG uptake can mimic metastatic disease. 187: 1124-1128
(12) AJR 2006; Iagaru A, Henderson R. PET/CT followup in nonossifying fibroma. 187: 830-832
(13) Radiology 2007; Hetts SW, et al. Case 110: Nonossifying fibroma. 243: 288-292
(14) Radiology 2007; Tateishi U, et al. Bone and soft-tissue sarcoma: preoperative staging with fluorine 18 fluorodeoxyglucose PET/CT and conventional imaging. 245: 839-847
(15) J Nucl Med 2007; Gerth HU, et al. Significant benefit of multimodality imaging: PET/CT compared with PET alone in staging and follow-up of patients with Ewing tumors: 48: 1932-1939
(16) Radiographics 2008; Zwaga T, et al. Osteosarcoma of the femur with skip, lymph node, and lung metastases. 28: 277-283
(17) J Nucl Med 2008; Benz MR, et al. Combined assessment of metabolic and volumetric changes for assessment of tumor response in patients with soft-ttisue sarcomas. 49: 1579-1584
(18) J Nucl Med 2008; Eary JF, et al. Spatial heterogeneity in sarcoma 18F-FDG uptake as a predictor of patient outcome. 49: 1973-1979
(19) J Nucl Med 2009; Costelloe CM, et al. 18F-FDG PET/CT as an indicator of progression-free and overall survival in osteosarcoma. 50: 340-347
(20) J Nucl Med 2009; Cheon GJ, et al. Prediction model of chemotherapy response in osteosarcoma by 18F-FDG PET and MRI. 50: 1435-1440
(21) J Nucl Med 2009; Dutour A, et al. 18F-FDG PET SUV max correlates with osteosarcoma histologic response to neoadjuvant chemotherapy: preclinical evaluation in an orthotopic rat model. 50: 1533-1540
(22) Radiographics 2009; Bestic JM, et al. Use of FDG PET in
staging, restaging, and assessment of therapy response in Ewing
sarcoma. 29: 1487-1501
(23) J Nucl Med 2011; Baum SH, et al. Contribution of PET/CT to
prediction of outcome in chidlren and young adults with
rhabdomyosarcoma. 52: 1535-1540
(24) J Nucl Med 2011; Eary JF, Conrad EU. Imaging in sarcoma. 52:
1903-1913
(25) Radiographics 2013; Murphey MD, et al. From the radiologic
pathology archives. Ewing sarcoma family of tumors:
radiologic-pathologic correlation. 33: 803-831
(26) AJR 2013; Rakheja R, et al. Necrosis on FDG PET/CT
correlates with prognosis and mortality in sarcomas. 201: 170-177
(27) J Nucl Med 2013; Byun BH, et al Combination of 18F-FDG
PET/CT
and diffusion-weighted MR imaging as a predictor of histologic
response to neoadjuvant chemotherapy: preliminary results in
osteosarcoma. 54: 1053-1059
(28) J Nucl Med 2013; Byun BH, et al. Initial metabolic tumor
volume meausred by 18F-FDG PET/CT can predict the
outcome of osteosarcoma of the extremities. 54: 1725-1732
(29) Radiology 2018; Im HJ, et al. Prognostic value of metabolic
and volumetric parameters of FDG PET in pediatric osteosarcoma: a
hypothesis-generated study. 287: 303-312