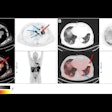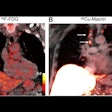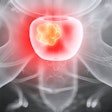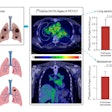
Sofie Biosciences has received Investigational New Drug (IND) clearance for a phase II clinical study of its investigational fibroblast activation protein inhibitor (FAPI) radiotracer in patients with gastrointestinal cancer.
The company will test F-18 FAPI-74 specifically in patients with gastric cancer, cholangiocarcinoma, hepatocellular carcinoma, pancreatic cancer, and colorectal cancer. The radiotracer is designed to bind to fibroblast activation protein (FAP), which is highly expressed across several tumor entities and has shown encouraging results in pre-clinical and clinical studies, Sophie said.