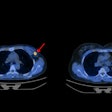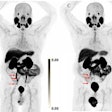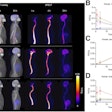
The U.S. Food and Drug Administration (FDA) has approved Blue Earth Diagnostics' F-18 rhPSMA-7.3 PET radiotracer for detecting prostate cancer.
F-18 rhPSMA-7.3 (Posluma) is indicated for men with prostate cancer with suspected metastasis who are candidates for initial definitive therapy or with suspected recurrence. The tracer is a radiohybrid agent consisting of a prostate-specific membrane antigen (PSMA) ligand that may be combined with isotopes such as F-18 for imaging purposes or lutetium-177 or actinium-225 for therapeutic use.
Posluma will be commercially available in early June 2023 and will become increasingly available nationally in coming months, the company said.