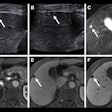
Optoacoustic imaging developer Seno Medical Instruments said it has finished enrolling patients in a study of its Imagio breast imaging system.
The aim of the study is to assess whether Imagio can help physicians determine if a woman could avoid unnecessary biopsies, Seno said. The results will serve as the basis for the firm's premarket approval (PMA) application with the U.S. Food and Drug Administration (FDA). More than 2,100 subjects have been enrolled at 16 U.S. institutions for the study, which will compare the sensitivity and specificity of Imagio with grayscale ultrasound imaging for breast lesions, according to Seno.
Dr. Reni Butler from Yale School of Medicine and Dr. Erin Neuschler from Northwestern University Feinberg School of Medicine are serving as co-principal investigators for the study.
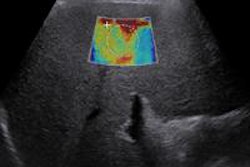
The Imagio system fuses optoacoustic energy with ultrasound to provide blood maps in and around suspicious breast masses. It is based on the phenomenon that cancerous tumors grow quickly and require significant amounts of blood and oxygen, the company said.