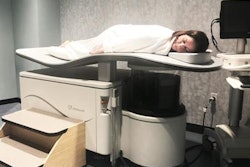
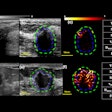
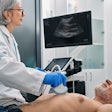
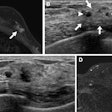
Ultrasound technology developer QT Ultrasound has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its Transmission Wave breast ultrasound scanner.
The transmission ultrasound technology is designed to generate high-fidelity 360° images of the breast, QT Ultrasound said. The company is currently conducting a clinical trial program to determine if transmission ultrasound technology can help reduce callback and false-positive biopsy rates, as well as replace screening mammography.