SAN FRANCISCO - A fully automated computer-aided diagnosis (CAD) system that provides differential diagnosis for breast calcifications may prove superior to neural network-based CAD systems, but must face down several challenges before it can move past the conceptual phase, according to its Finnish designers.
Janne Näppi from the Turku Centre for Computer Science in Finland presented the CAD system at the Computer Assisted Radiology and Surgery (CARS) meeting on June 29.
By estimating malignancy and pigeonholing calcification clusters with terms familiar to radiologists, the researchers believe that their system could provide a more detailed analysis than other CAD systems, which only indicate whether a cluster is malignant or benign. "Our system was designed to...produce the estimate of the classification -- whether it was malignant or benign -- and also an estimate of the characteristic patterns of the classifications," Näppi said.
Yet one major obstacle that the Finnish algorithm still needs to overcome is that it cannot determine if the calcifications were produced by cancer.
In their study sample, Näppi and his co-author, Peter Dean, sampled 70 cases associated with breast calcifications from the Turku University Central Hospital. Each case included a biopsy specimen that had been sliced into 5-mm sections. The sections were imaged separately at 14 kV. A radiologist then classified the calcification clusters with a system similar to the American College of Radiology's BI-RADS lexicon, Näppi said.
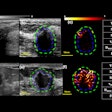
The areas of interest were digitized from the films to 1275 x 1023 images. The CAD system then read the images in stages: The preprocessing layer prepared the image for segmenting the calcifications; the segmentation layer located the calcifications; and the feature-analysis layer analyzed the calcification particles by size, shape, boundary, and contour. The computer assigned descriptions to the calcifications, such as "powderish," "granular," and "popcorn."
"We first looked at the major particles within the image. The major particles that are near to each other form a new cluster. Then the particles near to major particles form another cluster. The particles that are quite far from the major (sites) form their own cluster," Näppi said.
The system identified 748 clusters from 274 images. According to the results, the system and the radiologist made the correct diagnosis on 78% of the cases. In 65% of all clusters, the computer and the clinician were in sync; in 73% of these cases, the computer-assigned cluster label was deemed compatible with the radiologist’s classification, although there were some discrepancies.
In clusters that were non-powerderish, the computer and the radiologist identified the same lesions 43% of the time and were approximately similar in their findings in 81% of cases.
While the researchers were pleased with the system’s ability to differentiate between various types of breast calcifications, the system did fall short in several areas, Näppi said. The system cannot determine if the clusters are the result of cancer. In addition, there were cases of misclassification. Small clusters tended to suffer from segmentation errors, he said. In some instances, a benign-looking cluster appeared within a malignant case confirmed by histopathology. Finally, the quality of the digitized images is suboptimal at times.
According to a presentation the authors made at the Symposium for Computer Applications in Radiology in June, the major source for false-positives were film defects and regions of high-density parenchyma, which had a sharp contrast to the surrounding dark background. While it would be possible to identify and eliminate this problem, it also would lessen the system’s ability to detect subtle, but true, calcifications (Journal of Digital Imaging, May 2000, Vol.13:2, Suppl. 1, pp.130-132).
Still, this system is theoretically better than other CAD programs, the authors wrote in a paper published in the CARS book of proceedings.
"The system is conceptually superior to previously reported systems since the diagnosis is implemented in terms of explicit knowledge representation rather than a ‘black-box’ neural-network representation. The implementation allows detailed monitoring of the performance of subsystems in terms of differential diagnosis," they said (CARS 2000 Proceedings, pp.729-734).
CAD systems may be best used as a second opinion in order to "improve diagnostic accuracy and the consistency of radiologists' image interpretation," concluded researchers from the University of Chicago (European Journal of Radiology, Aug. 1999, Vol.31:2, pp.97-109).
By Shalmali Pal
AuntMinnie.com staff writer
June 30, 2000
Let AuntMinnie.com know what you think about this story.
Copyright © 2000 AuntMinnie.com