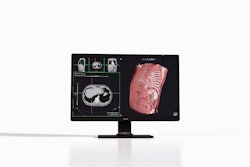
Avatar Medical has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for Avatar Medical Vision, a software platform for 3D medical image processing, review, and surgical planning using CT and MR imaging data.
The platform enables clinicians to interact with imaging data across standard operating room displays, glasses-free 3D systems, and virtual reality environments. It integrates with PACS and is designed for imaging-intensive specialties including neurosurgery, ENT, orthopedics, oncology, and interventional radiology, Avatar Medical said.
The clearance also establishes compatibility with the Barco Eonis 3D glasses-free display, forming the basis of a joint system called Eonis Vision. This allows clinicians and patients to view anatomy together in real time during consultations, according to the company.