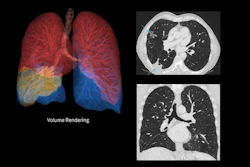
GE HealthCare (GEHC) has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for Photonova Spectra, a photon-counting computed tomography system powered by the company's Deep Silicon detector technology.
 GEHC's Photonova Spectra PCCT system
GEHC's Photonova Spectra PCCT system
The system features 8-bin energy resolution and wide detector coverage with a 0.23-second rotation speed, according to the company. Deep Silicon enables precise measurement of photon energy and supports material separation and characterization, allowing the system to distinguish between iodine, calcium, and fat, GEHC said. Photonova Spectra simultaneously captures spectral and ultrahigh-definition spatial data in a single scan without special setup or multiple protocols, the company noted.
The system incorporates NVIDIA accelerated computing technology and is designed to fit into existing GE HealthCare CT-ready rooms with minimal modification, according to the firm. The company is collaborating with University of Wisconsin-Madison and Stanford Medicine as the first U.S. clinical evaluation sites, it said.