A deep-learning AI model shows promise as a tool for opportunistic bone-health screening in children, according to a study published April 7 in Radiology.
A group at Seoul National University in South Korea developed the model using 1,464 chest x-rays paired with bone scans from pediatric patients and found it performed well at detecting low bone mineral density, noted Jae Won Choi, MD, and colleagues.
“Bone mineral density (BMD) measurement is essential for assessing bone health and monitoring bone mass accrual in children at risk for skeletal deficits,” the group wrote.
Clinicians are increasingly recognizing osteoporosis in children and adolescents with chronic conditions such as malignancies, inflammatory disorders, and neuromuscular impairments, the authors explained. While dual-energy x-ray absorptiometry (DEXA) scans are the gold standard for pediatric BMD measurement, it sees limited use in at-risk children because it typically requires a subspecialty referral, dedicated equipment, and specialist interpretation, they noted.
Recent advances in AI and deep learning have demonstrated potential, with studies in adults showing that chest x-ray–based models can estimate BMD. However, few studies have applied AI to chest x-rays for evaluating bone health in children and adolescents, according to the authors.
To bridge the gap, the group assembled a dataset of 1,464 radiograph-DEXA pairs from 1,188 patients younger than 18 years old (median age, 13 years; 26% with low BMD) at two tertiary hospitals. Most patients had hemato-oncologic diseases and inflammatory disorders and had undergone x-rays and DEXA scans within three months. Using a development set of 774 x-ray–DEXA pairs, the researchers trained a ResNet-50 convolutional neural network to predict BMD Z scores from the chest x-rays and clinical variables (age, sex, height, and weight).
In testing the model on an internal dataset of 376 pairs from 348 patients, the model achieved an area under the curve (AUC) of 0.92 for predicting BMD Z scores, with a sensitivity of 60% and a specificity of 95%. Next, on an external test set of 314 pairs, the model achieved an AUC of 0.90 for predicting BMD Z scores, with a sensitivity of 82% and a specificity of 85%.
For predicting low BMD -- defined as a Z score at or below −2.0 -- the model achieved an AUC of 0.92 internally and 0.90 in external validation, with sensitivity and specificity of 82% and 85%, respectively, the researchers reported.
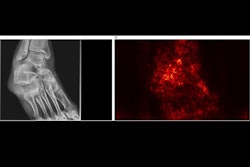
![Representative example of a 16-year-old male patient with underlying X-linked adrenoleukodystrophy. (A, B) Paired anteroposterior (AP) chest radiograph and dual-energy x-ray absorptiometry (DXA) report shows lumbar spine (L1 through L4) areal bone mineral density (BMD). The DXA report was reformatted for anonymization and improved readability. The patient had low BMD (Z score ≤ −2.0). (C) Model (chest radiography [CXR]–BMD) output shows the predicted raw BMD and Z score in comparison with the DXA reference standard, together with interpretability analyses using Shapley additive explanations (SHAP) and gradient-weighted class activation maps. The patient was classified as having low BMD, consistent with the reference standard. AM = age-matched, DEXA = dual-energy x-ray absorptiometry, RM2 = room 2, SNUH = Seoul National University Hospital, YA = young adult.](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/04/ai-children-bone-density.0snnf2EJjr.jpg?auto=format%2Ccompress&fit=max&q=70&w=400) Representative example of a 16-year-old male patient with underlying X-linked adrenoleukodystrophy. (A, B) Paired anteroposterior (AP) chest radiograph and dual-energy x-ray absorptiometry (DXA) report shows lumbar spine (L1 through L4) areal bone mineral density (BMD). The DXA report was reformatted for anonymization and improved readability. The patient had low BMD (Z score ≤ −2.0). (C) Model (chest radiography [CXR]–BMD) output shows the predicted raw BMD and Z score in comparison with the DXA reference standard, together with interpretability analyses using Shapley additive explanations (SHAP) and gradient-weighted class activation maps. The patient was classified as having low BMD, consistent with the reference standard. AM = age-matched, DEXA = dual-energy x-ray absorptiometry, RM2 = room 2, SNUH = Seoul National University Hospital, YA = young adult. RSNA
Representative example of a 16-year-old male patient with underlying X-linked adrenoleukodystrophy. (A, B) Paired anteroposterior (AP) chest radiograph and dual-energy x-ray absorptiometry (DXA) report shows lumbar spine (L1 through L4) areal bone mineral density (BMD). The DXA report was reformatted for anonymization and improved readability. The patient had low BMD (Z score ≤ −2.0). (C) Model (chest radiography [CXR]–BMD) output shows the predicted raw BMD and Z score in comparison with the DXA reference standard, together with interpretability analyses using Shapley additive explanations (SHAP) and gradient-weighted class activation maps. The patient was classified as having low BMD, consistent with the reference standard. AM = age-matched, DEXA = dual-energy x-ray absorptiometry, RM2 = room 2, SNUH = Seoul National University Hospital, YA = young adult. RSNA
Significantly, while no deep-learning model is fully interpretable, gradient-weighted class activation maps showed that the model concentrated on the lumbar and upper thoracic spine, regions that correspond to established radiographic markers of osteopenia, the group added.
As for study limitations, the researchers noted that the retrospective design precluded the evaluation of clinical outcomes such as fracture incidence or the impact of AI-guided screening on patient management. Also, longitudinal changes within individual patients, such as changes in BMD trajectories over time or treatment-related effects, were not assessed.
“In the future, rigorously designed prospective studies are needed to establish its clinical utility and to support translation into practice through multicenter validation, workflow integration, and compliance with medical-device and AI-specific regulatory requirements,” the group concluded.
The full study is available here.