ABK Biomedical is partnering with COR Development to build a dedicated manufacturing facility in Ashland, MO, for its Eye90 microspheres.
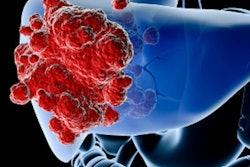
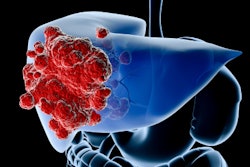
Eye90 microspheres are an yttrium-90 (Y-90) radioembolization device in clinical development for liver cancer treatment. The facility, to be constructed by Coil Construction at Cartwright Business Park, will serve as ABK's commercial-scale production site for Eye90 as the company advances toward full commercialization, according to an announcement. A key factor in the site selection was its proximity to the University of Missouri Research Reactor, the only U.S. producer of the Y-90 radioisotope used in the device, the company said.
Eye90 is the subject of a U.S. Food and Drug Administration (FDA) breakthrough device designation and is designed to enable multimodality imaging following implantation, allowing physicians to assess microsphere distribution after the procedure, ABK said.