A prostate cancer biopsy protocol based on targeted MRI rather than systemic ultrasound shows promise for reducing overdiagnosis by half, according to study results published December 8 in the New England Journal of Medicine.
"[The targeted MRI biopsy] strategy is sharply reducing the number of people who need to undergo tissue sampling, which is an unpleasant procedure with an associated risk of infection," study lead author Dr. Jonas Hugosson, PhD, of the University of Gothenburg said in a statement released by the university. "What's more, the strategy halves the risk of detecting a harmless tumor, which has been the biggest obstacle to introducing general screening for prostate cancer."
Many prostate cancers found on screening aren't clinically significant, which can lead to unnecessary treatment and thus increased risk of complications such as urinary incontinence and impotence, the group noted. Hugosson's team sought to assess the compare the performance of MRI-targeted biopsy for men with elevated prostate-specific antigen (PSA) levels and suspicious MRI results to systemic, ultrasound-guided biopsy.
The group conducted a study that included 17,980 men between the ages of 50 and 60 who underwent PSA screening. Those who had a PSA level of 3 ng per ml or higher then had an MRI exam of the prostate; a third of this group underwent systemic biopsy as well as MRI-targeted biopsy of any suspicious lesions found on MRI (reference group), while the remainder underwent MRI-targeted biopsy only (experimental group).
In the experimental group, 0.6% of patients were diagnosed with clinically insignificant prostate cancer, compared with 1.2% in the reference group -- a 50% reduction, Hugosson and colleagues noted. The team also found that relative risk of clinically significant prostate cancer in the experimental group was 0.81, compared with 0.6 in the reference group. Clinically significant cancer found only on systemic biopsy was diagnosed in 10 patients in the reference group, and all of these cases were intermediate risk, the team noted.
"The results show that the risk of finding an indolent (harmless) cancer was halved in the experimental group, while serious, potentially deadly cancers were found on an almost equal scale in both groups," the university said in its statement.
The study results suggest that MRI-target biopsy for screening and early detection of prostate cancer in men with elevated PSA and suspicious MRI results is a more tailored diagnostic option, according to the researchers.
"We must get away from the blind sampling of tissues that's still standard today, rely on the MRI examination, and thus switch diagnostics to taking samples only in those men in whom MRI has depicted tumors -- and then only do targeted samples in the area involved," Hugosson et al wrote.




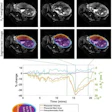
![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=100&q=70&w=100)





![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)