Researchers in Germany have developed a two-step deep-learning (DL) method for brain tumor MR imaging that predicts contrast enhancement without gadolinium, according to a presentation on May 11 at the ISMRM meeting in Cape Town, South Africa.
In addition, the two-step pipeline that incorporates four quantitative parameters, instead of two, outperforms single-step models, noted Dennis Thomas, PhD, a medical scientist in brain MRI at the Cooperative Brain Imaging Center and Institute of Neuroradiology at the Goethe University-affiliated University Hospital Frankfurt.
"Quantitative MRI enables better contrast enhancement prediction, and the inclusion of two other maps -- the quantitative susceptibility map and proton density map -- is making the accuracy better," Thomas explained, highlighting his group's goal of using quantitative MRI to predict contrast enhancement and reduce gadolinium-induced injury.
In brain tumor imaging, quantitative maps are useful for encoding tissue changes which correlate with blood brain barrier damage, essentially predicting contrast enhancement that would typically be observed through qualitative MRI or weighted imaging, Thomas said.
Gadolinium-based contrast agents (GBCAs) enable contrast- enhancement in MRI, but pose risks of toxicity and tissue retention. Predicting CE without GBCAs using quantitative MRI (qMRI) would offer a safer alternative, Thomas noted.
To evaluate CE prediction from qMRI, the group employed a standard multiparametric qMRI protocol for brain tumor assessment, using a variable flip-angle approach with an acquisition time of 12 minutes to measure four different quantitative parameters: T1, water content, T2 star, and quantitative susceptibility map (QSM).
A total of 46 brain tumor qMRI datasets were used to train a nnU-Net framework. The researchers created a qMRI model with two parameters (T1, T2*) and one with all four.
Thomas and colleagues used a five-fold cross-validation strategy. During each iteration, four folds (about 39 datasets) were used for model training, while the remaining fold (seven datasets) was reserved for validation. This procedure ensured that each dataset was used once for validation and four times for training, thereby reducing the risk of overfitting, according to Thomas.
For the tumors that were classified as contrast-enhancing tumors, researchers moved to the second step of the CE prediction model to compare the approaches.
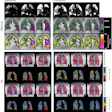
According to the findings, the four-parameter model achieved slightly higher Dice scores (0.60 ± 0.15) than the two-parameter model (0.52 ± 0.21), indicating improved CE prediction, according to Thomas.
 Performance of the qMRI-4-parameters model in five test subjects (P1–P5) with contrast enhancement. The corresponding Dice scores (DS) are shown on the left. CE: contrast-enhanced.Courtesy of ISMRM and Dennis Thomas, PhD
Performance of the qMRI-4-parameters model in five test subjects (P1–P5) with contrast enhancement. The corresponding Dice scores (DS) are shown on the left. CE: contrast-enhanced.Courtesy of ISMRM and Dennis Thomas, PhD
"The goal is hardware independent and physically meaningful maps," Thomas said. "When we use multiple vendors and multiple scanners, there is a decreased Dice score and a decreased specificity. ... Sequence first classification and then segmentation gives us better results."
The number of nonenhancing tumors in the training datasets is important for future research, Thomas said. While brain datasets without discernible lesions were correctly predicted in his study, nonenhancing tumors resulted in false positives, likely because no nonenhancing tumor datasets were included in training, according to Thomas. Additional nonenhancing tumor datasets are needed to improve specificity in such cases, he said.
Moreover, combining weighted images with qMRI maps may further enhance CE prediction accuracy, and future work should evaluate CE prediction performance using integrated weighted and qMRI data, Thomas noted.
The study only focused on a small dataset. However, the larger Raccoon AI Brain Tumor Project at 12 centers in Germany is expected to increase quantitative MRI datasets, Thomas explained. This will further support classification tasks, prediction tasks, and segmentation tasks, he said.
What's next? The group plans a side-by-side comparison of each of the four contrasts to determine the highest correlations.
Check out AuntMinnie's full coverage of ISMRM 2026 on our ShowCast.