Pulmonary Embolism
The PIOPED study was a Prospective Investigation of Pulmonary Embolism Diagnosis that tested utility of V/Q scanning. Subsequently, PIOPED II and the Prospective Investigative Study of Pulmonary Embolism Diagnosis have also been preformed and have aided in structuring the interpretation and reporting of the findings on the V/Q exam (see discussion below).
All patients enrolled in the original PIOPED study were to have a V/Q scan and a pulmonary arteriogram. Unfortunately, a large number of patients with normal or low probability scans who were enrolled in the study did not undergo arteriograms as many physicians did not want to have their patients exposed to the risks of the procedure in light of the V/Q scan findings. Because of this, there was a selection bias towards patients with intermediate or high probability scans (prevalence of PE based upon arteriogram for patients enrolled in the study was 33%). Such a bias would tend to overestimate the sensitivity, but underestimate specificity of V/Q scanning. Agreement among the readers was excellent for high, very low, and normal scans (92-95%), but agreement for intermediate and low probability scans was not as good [15]. In patients with chronic obstructive pulmonary disease, the sensitivity of a high probability V/Q scan is significantly lower compared to patients with no pre-existing cardiopulmonary disease. In fact, as the complexity of the patients underlying cardiopulmonary disease increases, so does the likelihood that the scan will not be informative (intermediate probability) [15]. Using PIOPED criteria, intermediate probability V/Q scans occurred in 60% of patients with COPD, but in only 13% of patients with normal CXR's [15].
An analysis of the regional distribution of PE on angiography demonstrated that PE occurred more frequently in the right lung, compared with the left, and more frequently in the lower lung zones compared with the middle or upper zones.
Clinical Findings
No single, or combination of, clinical findings is either specific or sensitive enough to diagnose or exclude PE. Symptoms of pulmonary embolism include tachypnea (most common), tachycardia, hypoxia, pleuritic chest pain, hemoptysis, and atrial fibrillation. Massive PE may be associated with cor pulmonale and the ECG may show right axis deviation, P-pulmonale, RBBB, or other evidence of right heart strain. A normal arterial blood gas does not exclude the presence of a PE. Although PE occurs most commonly from deep venous thrombosis in the lower extremity, about 10% arise from clot in the upper extremity primarily associated with an indwelling catheter.
Risk Factors
In the PIOPED study, 92% of the patients with pulmonary embolism
had at least one of the following risk factors [R]: immobilization, recent
surgery, underlying malignancy, history of deep venous
thrombosis or pulmonary embolism, estrogen use, pre-existing
cardiac disease. In patients without prior cardiac or
pulmonary disease only immobilization and surgery are significant
discriminating predisposing factors.
| Predisposing Factors: Patients with no previous cardiac or pulmonary disease [R] |
||
|
|
PE (n=117) |
No PE (n=248) |
|---|---|---|
| Immobilization | 66 (56%) | 81 (33%) * |
| Surgery | 63 (54%) | 78 (31%) * |
| Malignancy | 27 (23%) | 38 (15%) |
| Thrombophlebitis, Ever | 16 (14%) | 19 (8%) |
| Trauma - Lower Extremities | 12 (10%) | 25 (10%) |
| Estrogen Use | 10 (9%) | 26 (10%) |
| Stroke | 8 (7%) | 9 (4%) |
| Postpartum within past 3 mo. | 5 (4%) | 8 (3%) |
| * p < 0.001 | ||
Laboratory Findings:
None are suficiently diagnostic of PE for clinical use. However, D-dimer measurements of less than 500 micrograms/L could be used reliably to exclude PE in patients with an abnormal but not high-probability (inconclusive) lung scan [R]. On the other hand, normal oxigenation is not reliable to exclude PE [R].
Radiographic Findings:
CXR
The most common radiographic finding in patients with pulmonary embolism is atelectasis and/or parenchymal opacity. When looked for in the entire PIOPED population, it is found in at least 51% of patients with PE [R]. In a sub population of patients without prior cardiac or pulmonary disease, this finding occurred in 68% of those with PE and 48% of those without PE [R]. Pleural effusions within the affected hemithorax occurred in about 35% of patients with pulmonary embolism in the PIOPED study [R]. The majority of these pleural effusions were small, producing only costophrenic angle blunting. Occasionally, plain films may demonstrate findings such as Westermark's sign (local pulmonary oligemia, 11% of hemithoraces with PE), Fleischner's sign (regional oligemia in the presence of an ipsilateral enlarged pulmonary artery), and Hampton's Hump (a wedge-shaped pleural based density, 23% of hemithoraces with PE) [R].
|
CXR Findings in PIOPED Patients [R] |
|||
|
|
All pts |
Pts with PE |
Pts without PE |
|---|---|---|---|
|
Fleischner Sign |
216 (20.3%) |
77 (20.1%) |
139 (20.5%) |
|
Pulmonary Edema |
176 (16.6%) |
38 (10%) |
138 (20%) |
|
Normal |
166 (15.6%) |
45 (12%) |
121 (18%) |
|
Enlarged Hilum |
68 (6.4%) |
26 (7%) |
42 (6%) |
|
Enlarged Mediastinum |
50 (4.7%) |
18 (4.7%) |
32 (4.7%) |
|
COPD |
41 (3.9%) |
13 (3.4%) |
28 (4.1%) |
Helical CT (See discussion of PE in Chest Review Manual)
Angiography
Although considered the gold standard, angiography may not always detect the presence of emboli. Some indirect angiographic evidence for the presence of emboli is also non-specific. V/Q scans can provide an important "road map" to angiographers. Up to 95% of patients have been shown to have a PE ipsilateral to the abnormal side identified on V/Q scanning [1]. However, if the abnormally perfused segment on the V/Q scan appears normal at angiography, complete evaluation of the remainder of the lungs for the presence of pulmonary emboli is warranted as about 5% of patients can have isolated contralateral embolism [1].
Scintigraphic Examination
On V/Q scanning, a pulmonary embolism is characterized by a ventilation-perfusion mismatch: An area of normal ventilation corresponding to a segmental wedge-shaped area of decreased or absent perfusion which extends to the surface of the lung. Although PE typically has little or no effect on ventilation, if the scan is performed soon after a large (lobar) embolism has occurred (within 4 to 6 hours), rarely the vascular insult may induce reflex bronchoconstriction producing a matched ventilation-perfusion defect on both gas and aerosol ventilation studies [10].
In experimental animals, only 1.5% of emboli induced bronchoconstriction which lasted only a few hours. Bronchoconstriction is likely secondary to local hypocapnia (decreased carbon dioxide tension) and the release of serotonin, histamine, and other substances from platelets at the site of the embolus and it is a transient phenomena. Bronchoconstriction should be considered when multiple matched perfusion defects are seen in a patient without a history of airway disease and there has been only a short interval since the onset of symptoms. Follow-up ventilation study in these patients in 18 to 24 hours will demonstrate resolution of the ventilation abnormalities.
A NORMAL ventilation-perfusion scan essentially excludes the possibility of recent significant pulmonary embolus. The false-positive rate of high probability scans is only about 10%. In general, emboli are more frequent in the lower lobes due to the greater blood flow. Emboli are also frequently multiple (in 90%) and bilateral (in 85% of cases). Thus, unilateral decreased/absent perfusion to one lung is uncommonly the result of pulmonary embolism.
Injection of Tc-99m MAA into the veins of the feet may help to reveal a DVT. Venous obstruction or collateral flow can be seen. Images recorded several minutes after the injection may also show "hot spots" where the Tc-99m MAA is adherent to thrombus in the vein.
Interpretation is based on determining number of defects and descriptors of each one (see below).
Defect Descriptors, as Used in PIOPED:
V/Q ratio: Perfusion defect that has any ventilatory abnormality of comparable size is called a V/Q Match. If the perfusion defect has no corresponding ventilatory abnormality, or if it is either much more sever or larger than ventilatory abnormality, it is considered a V/Q Mismatch. NOTE: Any degree of perfusion decrement (NOT only the ones with completely absent perfusion) should be considered a perfusion defect. It is presumed that a partially occluding embolus could create a perfusion defect with perfusion that was diminished, but not absent. In fact, the pulmonary artery must be narrowed by at least 90% of its normal diameter before perfusion distal to the obstruction is reduced.
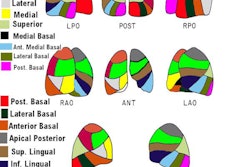
Size: Determining the size of a defect can be difficult. A knowledge of segmental anatomy is essential, and use of a schematic chart is encouraged to increase reading reproducibility and interobserver agreement (R). Note that, in general, the actual size of a perfusion defect is UNDER estimated by exam interpreters [13]- most likely due to shine through of radioactivity in contiguous segments [16]. In one study, only 44% of segmental defects were correctly identified as being greater than 75% of a segment [13]. Also- perfusion defects in the medial basal segment of the right lower lobe are often not detectable in any view [13]. Interobserver agreement regarding defect size is poor (about 40%) and even intraobserver agreement is only moderate (about 56%) [15].
* Small defect (small subsegmental): Less than 25% of a segment.
* Moderate defect (moderate subsegmental): > 25%, but < 75% of a segment.
* Large defect (segmental): Greater than 75% of a segment.
Vascular segmental distribution: A segmental defect
(independent of size) is a perfusion abnormality which may be
triangular or rectangular shaped, periphery based, and specially
located within one or several vascular segments. Nonsegmental
defects do not conform to segmental vascular anatomy and unlikely
to represent PE. Because the term "segmental" is also used in
sizing of defects, we prefer to call defect vascular
or nonvascular in appearance and distribution.
Reverse mismatch: A
reverse-mismatch occurs when the ventilation abnormality (absent
or minimal activity on wash-in images, with or without retention
of activity on washout) is worse than the perfusion abnormality
(ie: ventilation defect with preserved perfusion) [28]. This
finding can be seen in associaiton with mucus plugging of the
airways or atelectasis [28]. A reverse-mismatch is associated with
a functional right-to-left shunt in the pulmonary circulation
which may cause or contribute to hypoxemia [28].
Defect Descriptors, Identified in Studies Other Than PIOPED:
"Segmental Equivalent" Sizing: The moderate segmental
defect is counted as 0.5 of a large segmental defect (two moderate
sized segemental defects are therefore equivalent to one large
segmental defect). All moderate and large defects can be than
added up to result in total segmental equivalent units. (Example:
3 moderate segmental defects and 1 large = 2.5 segmental
equivalents). However, to simplify exam interpretation, some
authors have suggested equating a moderate to a large perfusion
defect as both have similar PPVs [32].
"Stripe Sign": A thin line (stripe) of activity (perfusion) between a perfusion defect and the adjacent pleural surface (best seen on a tangential view). The finding is associated with underlying emphysema and is likely related to spared perfusion in the cortex of the lung [6]. Iin the PIOPED study only 7% with a stripe sign actually had a pulmonary embolism corresponding to that segment [6]. Hence, in the absence of other perfusion abnormalities, the finding is considered very low probability for PE [20]. In cases in which there is a high clinical concern for pulmonary embolism, SPECT images may help to better define the defect [R].
"Triple Match": A matching perfusion, ventilation, and CXR abnormality is referred to as a "triple match". [8]. The overall prevalence of PE in all lung zones with triple matches was 26% in the PIOPED population [8]. There were no significant differences between the size of the matching V/Q defects and chest radiographic opacities and the prevalence of PE [8]. The prevalence of PE in small regions (less than 25% of a zone) with triple matches was 27%, compared with 21% in large regions (over 75% of a zone) with triple matches [8]. However, there was a difference in prevalence of PE between lung zones- pulmonary embolism was significantly more common in lower lung zone triple matches (upper - 11%; middle - 12%; lower - 33%) [3,8]. Pulmonary embolism was significantly more common in lower lung zone triple matches [8]. Therefore, a triple-match in the upper or middle lung zones is considered low probability for PE, but a triple-match in the lower lung zone should be interpreted as intermediate probability [8].
Interpretation Criteria
Interpretation is based on fulfillment of specified criteria. Original intrepretation criteria were Biello's and PIOPED (the original PIOPED criteria was tested prospectively). Modifications to PIOPED criteria have been suggested on the bases of retrospective PIOPED data review and were used for the PIOPED II study. Interobserver variability in scan interpretations is good when using the PIOPED criteria [18]. The new criteria shown below were used for the PIOPED II trial [19].
- The use of a new classification scheme has been suggested in
order to decrease the rate of non-diagnostic interpretations.
Gottschalk suggests that that a high probability study be
interpreted as "PE present" and that normal and very low
probability exams be interpreted as "PE absent" for a more
conclusive interpretation [20]. All other exams should be
interpreted as "PE uncertain" [20]. The Modified PIOPED II
criteria and a Trinary
System have been proposed using this type of scheme
[22].
In PIOPED I, 76.6% of scans were non-diagnostic (32.6% low
probability and 44% intermediate probability) [22]. In PIOPED II,
73.5% of patients had scans had a definitive interpretation (i.e.:
could be interpreted as PE present or PE absent) [21,22]. The
sensitivity for a high probability (PE present) exam in PIOPED II
was 77.4% (85.1% with CTA, but without DSA results) with a PPV of
87.2%, while the specificity of PE absent exams (very low or
normal) was 97.7% (98.2% with CTA, but without DSA results) [21].
Thus- V/Q imaging could provide a definitive diagnosis in a
majority of patients [21].
Other authors feel that normal, very low probability, and low
probability exams should be classified as "negative" (less than
10% likelihood for PE) with an acceptable false negative rate of
only 1.2% at followup [26,27]. These authors also suggest that
patients with greater than 1 segmental mismatch and no underlying
cardiopulmonary disease should also be considered in the "high
proability" or "PE present" category (PPV 86%) [27]. However, the
authors also point out that if there is a discordance between the
V/Q result and the objective clinical assessment, further testing
may be required [27].
Another criteria set has also been proposed by the Prospective
Investigative Study of Pulmonary Embolism Diagnosis (PISAPED Trial) using the
perfusion scan and the chest radiograph for evaluation [22]. This
criteria has been suggested to have a higher positive predictive
value and fewer not diagnostic interpretations [22].
Modified PIOPED II
High Probability (80-100% likelihood for PE [12,19]):
1. At least 2 large mismatched perfusion defects substantially larger than corresponding ventilation or x-ray abnormalities, or without any ventilation or x-ray abnormalities, or the arithmetic equivalent in moderate or large and moderate defects (two moderate defects = one large defect). A high probability lung scan confirms a very high likelihood for pulmonary embolism and justifies treatment with anticoagulation (unless contraindicated) [18].
Caveat - It has been suggested that 2.5 mismatched large segmental defects (or the arithmetic equivalent) is a better threshold for calling a scan high probability, as it associated with a 100% probability of PE in the PIOPED population [12].
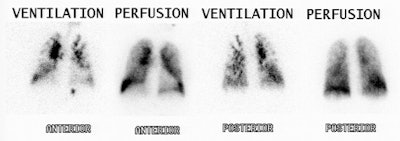
|
High probability scan for pulmonary embolism: The patient shown in the case below had undergone right knee prosthesis surgery and had a known lower extremity deep venous thrombosis. The patient had become acutely short of breath and a V/Q scan was ordered to evaluate for the presence of pulmonary embolism. The perfusion scan demonstrated multiple large and moderate sized segmental perfusion defects within both lungs- consistent with "showering" emboli. |
|
|
Intermediate Probability (20-80% likelihood for PE [12,19]):
1. 0.5-1.5 large mismatched perfusion defects or the arithmetic equivalent in moderate or large and moderate defects.
2. Solitary moderate or large segmental triple-match in the lower lobe.
3. Multiple opacities with associated perfusion defects.
4. Difficult to categorize as high or low, or not described as low or high. .
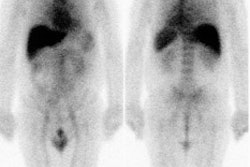
|
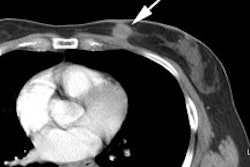
Intermediate probability scan for pulmonary embolism: The patient shown below had a single large segmental perfusion defect in the superior ligular segment of the left upper lobe. Further evaluation was recommended to exclude a pulmonary embolism. A CT PE study confirmed the presence of a PE in this segment. No other emboli were identified. |
|
|
Low Probability (10-19% likelihood for PE [12,19]):
1. A single large or moderate matched segmental defect.
2. More than 3 small segmental perfusion defects with a normal CXR.
3. Moderate sized pleural effusion (larger than the CP angle, but less than 1/3 of the pleural cavity) with no other perfusion defects in either lung.
4. Markedly heterogenenous perfusion.
5. Probable PE mimic- absent perfusion in entire lung, solitary lobar mismatch, mass, or other CXR lesions causing mis-matches.
|
Low probability scan for pulmonary embolism: The patient had a clear CXR and complained of SOB. The ventilation portion of the exam demonstrates markedly heterogeneous ventilation with central deposition of aerosolized Tc-DTPA consistent with COPD. There is a matching perfusion pattern. Extensive matched V/Q abnormalities are appropriate for low probability, provided that the CXR is clear. |
|
|
Very Low Probability (less than 10%) [19,20]:
1. Non-segmental perfusion abnormalities (prominent hilum, cardiomegaly, elevated diaphragm, linear atelectasis, CP angle effusion) with no other perfusion defects in either lung
2. Perfusion defect smaller than CXR abnormality.
3. At least two V/Q matched defects with regionally normal CXR and some areas of normal perfusion elsewhere in the lungs.
4. One to three small segmental defects (less than 25% of a segment).
5. Solitary triple-matched defect in the mid or upper lung zone confined to a single segment.
6. Stripe sign present around the perfusion defect.
7. Perfusion defect from pleural effusion at least 1/3 of the pleural cavity with no other perfusion defect in either lung.
Normal:
1. No perfusion defects or perfusion exactly outlines the shape of the lungs seen on the chest radiograph (note that hila and aortic impressions may be seen and the chest radiograph and/or ventilation study may be abnormal).
V/Q Scan Interpretation Nuances:
"Gestalt Interpretation" is formed by an experienced reader based on detailed knowledge of the various published lung image interpretation algorithms, as well as clinical data, ancillary findings, and pathophysiological features of PE, which are integrated with the individual case presentation [17]. A gestalt interpretation can be more accurate than any individual criteria set [R]. The Gestalt interpretation takes into consideration the fact that among patients with no underlying cardiopulmonary disease, fewer mismatched segmental defects are required to give a particular specificity and positive predictive value [15]. For instance, a previously healthy 25 year old female on birth control pills with a normal CXR and a single large segmental ventilation-perfusion mismatch in the lower lung zones on V/Q scanning may be interpreted to have a high probability study by an experienced reader. The Gestalt interpretation has been shown to have good-to-excellent intra- and interobserver variability [17,18].
A single moderate sized V/Q mismatch would qualify for low probability in the original PIOPED schema. However, such a finding was found to harbor PE in 36% of cases in the PIOPED cases [12]. Hence, it has since been considered to indicate intermediate probability.
A single matched (any size) V/Q defect has a 26% incidence of PE in the PIOPED population, hence, it is suggested to be classified as intermediate probability [12].
Defects associated with pleural effusion: Ipsilateral pleural effusions can be found in up to 35% of patients with acute pulmonary embolism. In the PIOPED study, the majority of patients with PE and pleural effusions had small effusions which caused blunting of the costophrenic angle. When the pleural effusion caused an isolated perfusion defect congruent with the size of the pleural effusion, and when the pleural effusion was limited to the costophrenic angle, the incidence of PE was 25% [12]. Therefore, matching V/Q defects with a small effusion represent intermediate probability for PE. In patients with no prior cardiopulmonary disease and an effusion that was greater or equal to one-third of the hemithorax, no PE was found, (i.e., low probability) [3,R]. However, some authors outside the PIOPED group argue that any size perfusion defect should be considered intermediate probability in association with any size effusion [R].
Generalized CXR abnormality: A generalized abnormality on CXR (such as diffuse pulmonary edema or reticulonodular disease) may not cause the perfusion lung scan to be abnormal- in fact, up to 73% of patients with such findings can have normal or near normal perfusion images [15].
Clinical Probability of PE:
The actual incidence of PE is highly dependent upon the pretest (clinical) likelihood for the presence of pulmonary embolism. The utility of the V/Q is optimized when it is interpreted in conjunction with the clinical likelihood for PE. This was well demonstrated in the PIOPED [R]:
Clinical Science Probability |
||||
| Scan Probability | 80 - 100% | 20 - 79% | 0 - 19% | All Probabilities |
| High | 96% | 88% | 56% | 87% |
| Intermediate | 66% | 28% | 16% | 30% |
| Low | 40% | 16% | 4% | 14% |
| Near normal/ normal | 0% | 6% | 2% | 4% |
| Total | 68% | 30% | 9% | 28% |
SPECT imaging in the evaluation of pulmonary embolism:
SPECT imaging has also been employed for the diagnosis of
pulmonary embolism [23]. The advantages of SPECT is its higher
contrast resolution and avoidance of over-lapping of small
perfusion defects by normal tissue (shine through from normal lung
segments can obscure small perfusion defects)- particularly
perfusion defects in the mediobasal segment of the lower lobe that
can be unnoticed on planar imaging [23,30]. In most studies, both
the sensitivity (80-100%) and specificity (93-100%) of SPECT
imaging for PE has been at least 90%, and the sensitivity,
specificity, and accuracy have generally been higher than that of
planar imaging [23,30]. False-negative SPECT exams have been
reported in 0.7-3.3% of cases [23]. Additionally, not all patients
with PE have a classic V/Q mismatch pattern because some will
develop pulmonary infarction that can result in a matched defect
[30].
For the exam, Technegas is a superior agent for high-quality
SPECT V/Q imaging as the distribution is more homogeneous
(particularly in patients with airways disease) due to its small
particle size (30-60 nm) and the agent remains fixed for the
duration of the study (unlike Tc-DTPA) [23,25,30]. The dose of
technegas required for the exam is about 500 MBq (13.5 mCi) [23].
The perfusion exam also requires a slightly larger dose of Tc-MAA
(mean activity 206-220 MBq) [23]. The larger doses will result in
a larger patient radiation exposure- approximately 1.5 mSv for the
ventilation exam and 3.4 mSv for the perfusion study (total 4.9
mSv), however, dose estimates of 2.5 mSv or less have also been
reported [23,30]. In comparison, 64-slice CTPE has an effective
dose of about 19.9 mSv [23]. If SPECT/CT is performed, a reduced
dose exam (20-80mA) is generally adequate and associated with an
additional radiaiton dose of 1-2 mSv [30]. To reduce
respiratory-motion misregistration, it has been recommended that
the CT scan be acquired during breath-hold at mid-inspiration, or
with the patient continuing shallow breathing [30].
Multidetector cameras are preferred for V/Q SPECT [30]. Total
acquisition time for the SPECT V/Q exam is between 20-30 minutes
for a dual headed camera [23]. A recommended SPECT acquisition is
3? steps over a 360? acquisition using 12 sec per step for the
ventilation exam and 8 sec per step for the perfusion study [30].
A low-energy, high resolution collimator and a matrix of 128 x 128
(although a 64 x 64 matrix can also be used) [30]. The images
should be reconstructed using ordered-subset expectation
maximization (8 iterations, 4 subsets) and post-reconstruction
filtering with a 3-dimensional Butterworth filter (cutoff 0.8
cycles/cm; order 9) [30].
For V/Q SPECT exam interpretation, the European Association of
Nuclear Medicine guidelines recommend that the exam be considered
positive for PE if there is one or more segmental or two or more
subsegmental mismatches that conform to the vascular anatomy
(sensitivity 92% and specificity 91%) [29,30,31]. In that same
study, the post-test probability of PE with a negative VQ SPECT
result, but a high clinical suspicion was 12% and the post test
probability with a positive exam, but a low clinical suspicion was
53% [29]. Probabilistic reporting as used for planar scanner is
not recommended and has not been validated for V/Q SPECT [30].
False positives
Patients with prior pulmonary embolism can have persistent ventilation-perfusion abnormalities [15]. Pulmonary emboli resolve because of natural thrombolytic processes [15]. A residual perfusion defect can be found in 9% to 30% of patients and incomplete resolution may be more common in patients with underlying cardiopulmonary disease [15].
Among young non-smokers approximately 7% may demonstrate subsegmental perfusion defects and 3-4% lobular or segmental defects. As many as 10% of smokers may exhibit some type of perfusion defect. Xenon washout studies have also demonstrated mild delay in regional clearance and heterogeneity of clearance time in young smokers. These changes become progressively more severe and irreversible in long-term smokers.
Multiple nonsegmental, matched ventilation-perfusion defects can be seen following smoking of free based cocaine and snorting heroin. This may be related to the local vasoconstrictive and bronchoconstrictive actions of cocaine [9].
Bronchoalveolar lavage can also produce perfusion defects and lung scanning should be delayed at least 24 hours following the procedure to avoid mis-interpretations [7].
Post-scan clinical decisions
Recommendations regarding the further evaluation of patients, based upon the V/Q scan findings and clinical likelihood of embolism: [Radiologic Clinics of North America, July 1993, p. 856]
* Patients with normal studies require no further evaluation or treatment.
* Patients with a low probability scan, a low clinical likelihood, and no clinical evidence of DVT do not require anticoagulation or further workup.
* Patients with a low probability scan, and an intermediate or high clinical suspicion of embolism, should have CTPA imaging or serial ultrasound of the lower extremities to exclude DVT. If these studies are negative, then anticoagulation is not necessary, however, careful follow-up is recommended. In general, complications from PE are identified infrequently in patients with low probability V/Q scans [4]. Three series have shown a less than 1% incidence of serious thromboembolic events over a 6 month or longer followup period after a low-probability V/Q exam [24].
* Patients with an intermediate probability scan require ultrasound of the lower extremities. If these studies are negative, further evaluation with angiography may be warranted. The low morbidity (6%) and mortality (0.5%) associated with pulmonary angiography justifies its use in selected patients with suspected pulmonary embolism [3].
Duplex US has a sensitivity of 95% and a specificity of 99% for the detection of proximal vein DVT. In one study, DVT was detected in 21% of patients with normal lung scans, 15% with low probability scans, 30% with intermediate prob. scans, and 50% of patients with high prob. scans. The presence of a DVT in patients with an intermediate probability lung scan raises the likelihood of PE to 93% [4].
On follow-up, a change in the perfusion pattern can be seen as
early as a few days following the event. Defects may become
smaller and disappear, or large clots may fragment creating new
peripheral defects. A scan performed as early as 5 to 10 days
post-event frequently shows marked resolution of perfusion defects
(except in the elderly, about 50% of defects will have resolved by
2 weeks in patients treated with heparin). Approximately 15% of
patients will have a persistent perfusion defect at one year post
event, although the abnormality is typically less well defined.
Resolution of a scan defect is generally age dependent, with about
50% of patients less than 40 years old having complete resolution
of defects, while few patients over the age of 60 years will have
their scan return to normal. Emboli which lead to pulmonary
infarction are also unlikely to have resolution of the perfusion
defect. A 4 week follow-up exam is beneficial to serve as a
baseline study should these patients become symptomatic in the
future. However, other authors recommend a one week and 6 month
followup scan [33]. Persistent perfusion defects on perfusion
scintigraphy 6 months after an index event is a risk factor for
recurrence and development of chronic post embolic pulmonary
hypertension [33]. The severity of PE at baseline can predict the
likelihood for complete perfusion recovery at 6 months- the less
severe the initial reduction in perfusion, the higher the
probability that a patient will have a normal perfusion scan at 6
months [33]. Patients may also acutely or chronically develop a
"hyperperfusion sign"- regions in which perfusion is markedly
increased in comparison to ventilation [33]. This is due to areas
of abnormally increased perfusion in the upper and anterior lungs
because of a pulmonary blood flow shift from the lower and
posterior lungs due to predominant embolic obstruction in these
regions [33]. This typically disappears after the first week of
therapy, but persistence of this sign correlates with elevated
pulmonary artery pressures [33].
Treatment for pulmonary embolism
Treatment for PE most commonly consists of anticoagulation with heparin or coumadin. When properly diagnosed and treated, death attributed to pulmonary embolism is relatively uncommon (occurring in about 2.5% of cases) [2]. Thrombolytic agents (such as urokinase or tPA) are not routinely used in the treatment of pulmonary embolism. Such treatment is generally reserved for patients with large clot burdens (i.e., occlusion of greater than 50% of the pulmonary bed, or 25-30% in patients with pre-existing cardiac or pulmonary disease) or for patients who are hemodynamically unstable.
REFERENCES:
(1) Radiology 1999; Davey NC, et al. Ventilation-perfusion lung
scintigraphy as a guide for pulmonary angiography in the
localization of pulmonary embolism. 213: 51-57
(2) N Engl J Med 1992; Carson JL, et al. The clinical course of pulmonary embolism. 326:1240-1245
(3) J Nucl Med 1995; Worsley DF, Alavi A. Comprehensive analysis of the results of the PIOPED Study. Prospective Investigation of Pulmonary Embolism Diagnosis Study. 36: 2380-2387
(4) Radiology 1994; Smith LL, et al. Pulmonary embolism: Confirmation with venous duplex US as adjunct to lung scanning. 191: 143-147
(5) Radiology 1994; Sostman HD, et al. Evaluation of revised criteria for ventilation-perfusion scintigraphy in patients with suspected pulmonary embolism. 193: 103-107
(6) Radiology 1992; Sostman HD, Gottschalk A. Prospective validation of the stripe sign in ventilation-perfusion scintigraphy. 184: 455-459
(7) J Nucl Med 1993; Chen CC, et al. Abnormalities on ventilation/perfusion lung scans induced by bronchoalveolar lavage. 34: 1854-1858
(8) J Nucl Med 1993; Worsley DF, et al. Detailed analysis of patients with matched ventilation-perfusion defects and chest radiographic opacities. 34: 1851-1853
(9) Semin Nucl Med 1993; Benson ML, Balseiro J. Multiple matched ventilation-perfusion defects in illicit drug use. 23: 180-183
(10) J Nucl Med 1994; Aug: 1351-57
(11) J Nucl Med 1993; Gottschalk A, et al. Ventilation-perfusion scintigraphy in the PIOPED study. Part I. Data collection and tabulation. 34: 1109-1118
(12) J Nucl Med 1993; Gottschalk A, et al. Ventilation-perfusion scintigraphy in the PIOPED study. Part II. Evaluation of the scintigraphic criteria and interpretations. 34: 1119-1126
(13) J Nucl Med 1992; Morrell NW, et al. The underestimation of segmental defect size in radionuclide lung scanning. 34: 370-374
(14) Semin Nucl Med 1991; Juni JE, Alavi A. Lung scanning in the diagnosis of pulmonary embolism: the emperor redressed. 21: 281-296
(15) Prog Cardiovasc Dis. 1994; Stein PD, Gottschalk A. Critical review of ventilation/perfusion lung scans in acute pulmonary embolism. 37: 13-24
(16) J Nucl Med 2002; Meignan MA. Lung ventilation/perfusion SPECT: the right technique for hard times. 43: 648-651
(17) J Nucl Med 2002; Hagen PJ, et al. How to use a Gestalt interpretation for ventilation-perfusion lung scintigraphy. 43: 1317-1323
(18) J Nucl Med 2003; Hagen PJ, et al. Comparison of observer variability and accuracy of different criteria for lung scan interpretation. 44: 739-744
(19) Respiration 2003; Schumichen C. V/Q-scanning /SPECT for the diagnosis of pulmonary embolism. 70: 329-342
(20) J Nucl Med 2007; Gottschalk A, et al. Very low probability interpretation of V/Q lung scans in combination with low probability objective clinical assessment reliability excludes pulmonary embolism. 48: 1411-1415
(21) Radiology 2008; Sostman HD, et al. Acute pulmonary embolism: sensivity and specificity of ventilation-perfusion scintigraphy in PIOPED II study. 246: 941-946
(22) J Nucl Med 2008; Sostman HD, et al. Sensitivity and specificity of perfusion scintigraphy combined with chest radiography for acute pulmonary embolism in PIOPED II. 49: 1741-1748
(23) J Nucl Med 2009; SPECT in acute pulmonary embolism. 50: 1999-2007
(24) AJR 2010; Stein EG, et al. Success of a safe and simple algorithm to reduce use of CT pulmonary angiography in the emergency department. 194: 392-397
(25) J Nucl Med 2010; Jogi J, et al Ventilation-perfusion SPECT
with 99mTc-DTPA versus technegas: a head-to-head study
in obstructive and nonobstructive disease. 51: 735-741
(26) J Nucl Med 2011; Sostman HD, Pistolesi M. Scintigraphy for
pulmonary embolism: too old to rock 'n' roll, too young to die?
52: 1505-1507
(27) J Nucl Med 2011; Glasser JE, et al. Successful and safe
implementation of a trinary interpretation and reporting strategy
for V/Q lung scintigraphy. 52: 1508-1512
(28) Radiographics 2013; Uliel L, et al. Nuclear medicine in the
acute clinical setting: indications, imaging findings, and
potential pitfalls. 33: 375-396
(29) J NUcl Med 2013; Le ROux PY, et al. V/Q SPECT interpretation
for pulmonary embolism diagnosis: which criteria to use? 54:
1077-1081
(30) J Nucl Med 2013; Roach PJ, et al. V/Q scanning using SPECT
and SPECT/CT. 54: 1588-1596
(31) J Nucl Med 2015; Le Roux PY, et al. Pulmonary scintigraphy
for the diagnosis of acute pulmonary embolism: a survey of current
practices in Australia, Canada, and France. 56: 1212-1217
(32) J Nucl Med 2017; Metter D, et al. Current status of
ventilation-perfusion scintigraphy for suspected pulmonary
embolism. 489-494
(33) J Nucl Med 2019; Marconi L, et al. Should perfusion
scintigraphy be performed to follow patients with acute pulmonary
embolism? If so, when? 60: 1134-1139