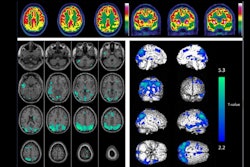
Patients with less tau pathology on PET scans may respond better to treatment with the new Alzheimer’s disease drug donanemab, according to an October 25 news report in the journal Practical Neurology.
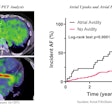
Researchers from donanemab developer Eli Lilly presented a follow-up analysis of data from the TRAILBLAZER-ALZ 2 clinical trial at the Clinical Trials on Alzheimer’s Disease (CTAD) 2023 conference in Boston. According to the findings, participants with baseline characteristics of early-stage Alzheimer’s disease, including lower tau PET, younger age, and plasma phosphorylated tau (p-tau217) levels, showed greater benefit when treated with donanemab.
Disease progression in the trial was assessed according to two measures: the Integrated Alzheimer Disease Rating Scale (iADRS) and Clinical Dementia Rating - Sum of Boxes (CDR-SB) scores of clinical decline over 76 weeks.
Specifically, patients treated with donanemab for 76 weeks who had low to medium tau PET brain pathology had a 35% (iADRS) and 36% (CDR-SB) slower clinical decline compared to patients in the placebo arm of the trial. Moreover, patients younger than 75 years old who had low to medium tau PET had a 48% (iADRS) and 45% (CDR-SB) slower decline compared with placebo patients.
Tau protein neurofibrillary tangles and beta-amyloid plaque deposits are the two major pathological hallmarks of Alzheimer's disease, with tau PET and amyloid PET scans used to diagnose the disease. Research suggests that the accumulation of these proteins is a continuous process that starts decades before the onset of symptoms.
Donanemab is a monoclonal antibody designed to bind to beta-amyloid protein that already has formed into plaque deposits. The drug then stimulates the immune system to attack these plaque deposits and clear them from the brain.
Ultimately, the reduced progression of the disease seen in these latest results may strengthen the hypothesis of an amyloid-mediated tauopathy. In an earlier study, Eli Lilly scientists suggested that a "parsimonious interpretation" is that early reduction of amyloid plaque may reduce the amount of time during which amyloid plaque can drive downstream tau pathology, and thus possibly prevent disease progression.
Donanemab is currently under review for approval by the U.S. Food and Drug Administration, with a decision expected by the end of 2023.
The full report is available here.