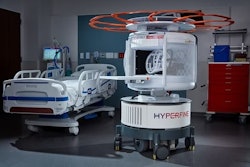
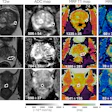
Hyperfine has received CE Marking and UK Conformity Assessment (UKCA) approval for its Swoop portable brain MRI system and the latest version of its Optive AI software.
These approvals make way for commercialization across Europe and the U.K.
The next-generation Swoop system, which received clearance from the U.S. Food and Drug Administration (FDA) in the second quarter of 2025, delivers improvements in image quality, workflow, and user experience compared with the previous generation, according to the company.
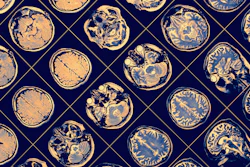
The accompanying Optive AI software, FDA-cleared in December 2025, includes a new multidirection diffusion-weighted imaging (DWI) sequence that acquires and averages images from multiple diffusion directions. The capability is designed to improve detection of small strokes and enhance performance in acute neurological care.
The Optive AI software is included in the next-generation Swoop system and is also available as a standalone upgrade for existing users, the company said.
Hyperfine plans to begin commercialization in Europe and the U.K. in the third quarter of 2026 through its existing distributor network across more than a dozen countries.