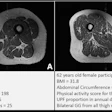
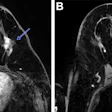
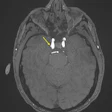
GE HealthCare (GEHC) has dosed the first patient in the phase II/III LUMINA trial of mangaciclanol, its investigational manganese-based MRI contrast agent, at Mayo Clinic in Rochester, MN.
Mangaciclanol has received fast-track designation from the U.S. Food and Drug Administration (FDA) for use in adults and pediatric patients ages two years and older for MRI to detect and visualize lesions with abnormal vascularity in the central nervous system and body. The agent is intended as an alternative to gadolinium-based contrast agents, the current standard of care, GE HealthCare said.
Manganese is an endogenous element naturally occurring in the body, and the macrocyclic structure of mangaciclanol is designed to reduce the possibility of retention, according to the firm. Manganese is also more widely available geographically than gadolinium, whose supply is largely dependent on Chinese mining and processing infrastructure, potentially reducing supply chain risk, GE HealthCare noted.