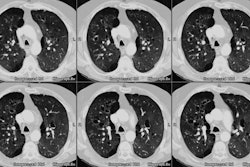
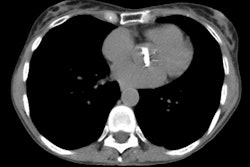
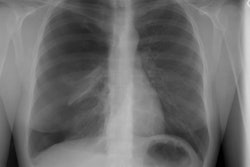
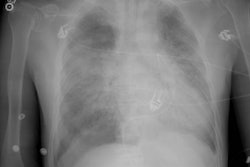
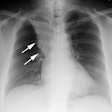
Bone Marrow (Hematopoietic stem cell) Transplant:
Clinical:
Bone marrow or hematopoietic stem cell transplantation (BMT) involves the intravenous infusion of hematopoietic progenitor cells to reestablish marrow function. Allogeneic transplantation refers to the transfer of marrow from a donor to a recipient who is not an identical twin, while autologous transplantation involves the use of the patients own marrow. Pulmonary complications occur in about half of allogeneic BMT patients and are a common cause of mortality (in about 30% of patients).Pulmonary complications can be classified as occuring during the neutropenic or pre-engraftment period (0 to 30 days following BMT), early or post-engraftment period (31 to 100 days following transplant), or late post-engraftment period (after 100 days) [13]. During the neutropenic period the transplanted marrow is not yet functioning and the patient remains neutropenic and the frequency of infectious and non-infectious pulmonary complications is similar [13]. Common neutropenic phase complications include fungal infection (especially with aspergillus), alveolar hemorrhage, pulmonary edema, and drug reactions.
During the early post transplant period (31-100 days) the neutrophil count is usually normal, but there is impaired cellular and humoral immunity [13]. Early post-engraftment period complications include CMV pneumonia. Non-infectious early post transplant complications include acute GVHD, acute radiaiton pneumonitis, and rarely pulmonary cytolytic thrombi [13].
Late phase complications include bronchiolitis obliterans, bronchiolitis obliterans and organizing pneumonia, chronic graft-versus-host disease, and nonclassifiable interstitial pneumonia [13]. Nearly one third of bone marrow transplant patients develop progressive interstitial pulmonary infiltrates during the first 6 months following the procedure
Infectious Complications:
Infection is the most common complication in BMT patients including bacterial, fungal (12-56% of pneumonias), and CMV. The incidence of pulmonary infection has been reported to be higher in allogenic transplants [7]. Occurrence of a pulmonary infection is associated with an increased mortality rate [7]. Specific infectious etiologies tend to occur during predictable times following transplant.The majority of fungal infections occur within the first 30 days after transplantation during the neutropenic phase before successful establishment of the graft [6,7,10]. Aspergillus is the most common fungal pathogen [6,7] accounting for 90% of fungal infections [10]. Nodules, masses, or nodules and masses are the most common radiographic finding- commonly involving the upper lung zones [7]. An area of ground-glass attenuation about the nodules can be seen and is referred to as the "CT halo sign" representing a rim of hemorrhagic infarction [8]. Cavitation may not occur [7].
CMV infection develops between 30 to 120 days after BMT and is seen in 50-70% of allogeneic recipients. CMV infection occurs much less commonly in autologous and syngeneic transplant patients [7]. Between 15% to 30% of these patients will develop a clinical pulmonary infection [5]. The infection is caused by reactivation of the latent virus at a time of profound immunosuppression or by the infusion of seropositive marrow into a previously seronegative patient [10]. Infusion of seropositive ceels into a seronegative recipient increases the risk of infection and clinical pneumonitis [5]. Mortality is high (80-85%) and uniformly fatal if not treated [5].Combination therapy with ganciclovir and immune globulin can result in survival rates between 35-75% [7]. The incidence of CMV infection is decreasing due to prophylactic treatment with ganciclovir. The radiographic findings in CMV infection are variable and range from a reticular pattern to air space consolidation. The findings are frequently bilateral and tend to involve the middle and low lung zones [7]. Small pleural effusions occur in about 20% of cases [7]. On CT the most common finding in CMV infection is small, 1 to 5 mm ill-defined nodules associated with areas of ground-glass attenuation [8].
PCP pneumonia occurs in less than 10% of BMT patients due to effective prophylaxis with trimethoprim-sulfamethoxazole [5]. When PCP infection does occur, it is generally rapidly progressive.
Bacterial pneumonias are the most common cause of pulmonary infection occurring more than 1 year after transplant [7]. Bacterial pneumonias are rare in bone marrow transplant patients during the pre-engraftment period due to the routine use of empiric antibiotics in neutropenic patients [7]. The most common organisms resulting in bacterial pneumonia before marrow engraftment are gram-negative bacteria from the GI tract and gram-positive organisms such as Staphylococcus and Streptococcus [10]. During the early phase after transplantation the most frequent causes of bacterial pneumonia are gram-negative organisms such as Pseudomonas aeruginosa and Escherichia coli [10]. Six months after bone marrow transplant, the most common causes of sinopulmonary infection are Streptococcus and Staphylococcus [10].
Viral pneumonia can be seen in up to 20% of patients and typically produces acute symptoms (fever, non-productive cough, dyspnea, and hypoxemia) [11]. Common causes of viral infection include influenza A, parainfluenza virus, respiratory syncytial virus, and human metapneumovirus [11]. CXR may not reveal parenchymal findings and HRCT is often used in these patients if there is a high clinical suspicion for infection (up to 50% of patients with normal CXR's can have findings on HRCT) [11]. Common CT findings in viral infection include areas of ground glass attenuation, ground-glass nodules, tree-in-bud changes, bronchovascular thickening, and areas of airspace consolidation [11].
Non-Infectious Complications:
Pulmonary Edema:
Pulmonary edema is the most common noninfectious post-BMT complication that is typically seen in 10 to 65% of patients 2 to 3 weeks following the procedure. Autologous and allogenic transplant recipients are affected equally [13]. The etiology is uncertain, but is likely multifactoral and related to hydrostatic causes and increased alveolar permeability [13]. Patients present with dyspnea, hypoxemia, and even fever. CXR will demonstrate interstitial pulmonary edema with Kerley lines, enlarged pulmonary vessels, and diffuse, bilateral ground-glass pulmonary opacities. Cardiomegaly is usually absent. Treatment is diuresis and resolution is usually slow- over 1 to 2 weeks.Engraftment syndrome:
Engraftment syndrome (PERDS) is a noninfectious form of diffuse
capillary
leak [9,13]. The syndrome occurs most frequently following
autologous BMT
(7-11% of cases) and is rarely seen following allogenic transplant
[9].
The median time of onset is 7-21 days following BMT (during the
engraftment of hematopoietic stems cells and neutrophil recovery)
[9,13]. The syndrome appears to be the result from the release of
proinflammatory cytokines during engraftment [13]. Patients
present
with fever, hypoxia, and may also have skin rash similar to that
seen
in acute graft-versus-host disease [9]. The prognosis is generally
good and the condition may either resolve spontaneously or respond
promptly to steroid treatment [13]. CXR findings are nonspecific
ranging from normal to bilateral airspace opacification with
effusions
[9]. CT scans can show bilateral ground glass opacification,
perihilar/peribronchial airspace consolidations smooth
interlobular septal thickening, and pleural effusions [9]. The
findings may be indistinguishable from pulmonary edema [13].
Idiopathic Pneumonia Syndrome:
This syndrome is defined as diffuse lung injury following BMT for which an infectious, cardiogenic, or iatrogenic (fluid overload) etiology is not identified. It is probably secondary to pulmonary toxicity related to chemotherapeutic and XRT regimens prior to transplantation. The syndrome is seen in about 10% (3-15%) of allogeneic BMT patients who undergo myeloablative regimensand usually occurs between 6 and 7 weeks following the procedure (the median time of onset is 20-42 days after transplant) [13]. Although IPS may also affect autologous BMT recipients, the incidence is lower, the onset is later, and the overall prognosis is better [13]. There is no recognized treatment. Mortality approaches 70% [9]. Patients present with rapidly progressive acute respiratory distress associated with cough, dyspnea, fever, and rales. The radiograph will demonstrate the presence of multilobar infiltrates.Diffuse Alveolar Hemorrhage:
Diffuse alveolar hemorrhage occurs in about 20% (5-30%) of BMT recipients. Some authors suggest it is more common in autologous recipients and is uncommon in allogeneic transplants, but other authors suggest a similar incidence in both groups [13]. It usually presents within the first two weeks (12-15 days [13]) following the procedure and its occurrence is associated with the onset of marrow recovery. Mortality is between 60 to 100% within 3 weeks of diagnosis [13], although high dose steroid therapy may improve survival. Predisposing conditions include patients with solid tumors, age over 40 years, renal insufficiency, radiation prior to BMT, and severe mucositis. Patients generally complain of dyspnea, non-productive cough, and fever [4]. Despite its name, hemoptysis is rare in the condition [4] (other authors report hemoptysis in 66% of cases [13]). A finding of more than 20% hemosiderin laden macrophages in BAL fluid is suggestive of DAH, however, the finding can also be seen in patients with low platelets or pulmonary hypertension [13]. Radiographic findings initially consist of bilateral reticular or patchy alveolar opacities usually in the mid to lower lung zones. Rapid progression to a severe, diffuse alveolar pattern is characteristic. On CT small areas of ground-glass attenuation confined to the secondary lobules can be seen.Drug Reaction:
Many of the induction agents used for BMT such as busulfan and methotrexate can produce a toxic pneumonitis. Such reactions can be found out to 12 weeks after BMT. Steroid therapy typically results in a rapid clinical improvement, usually over 1 to 5 days. Radiographic findings are non-specific and include bilateral diffuse, or patchy air-space opacities which may progress to diffuse consolidation.Bronchiolitis Obliterans:
See discussion below of graft-versus-host disease.Organizing Pneumonia:
Organizing pneumonia occurs within the first 3 months of transplant, and a second increased incidence is found in long term survivors of BMT. The incidence is low affecting 2-10% of patients [13]. It is more common in allogeneic BMT recipients [13]. Symptoms include a non-productive cough, dyspnea, and a low grade fever. Patients usually respond (about 80%) well to treatment with steroids. Findings include peripheral areas of ground-glass opacification or consolidation particularly in a sub-pleural and peribronchial distribution. Nodules, measuring 5 to 15 mm in size, often with well defined margins, may be seen and are more frequently observed in immunocompromised patients.Graft versus Host Disease:
Graft-versus-host disease results from an immune reaction of the immunocompetent donor cells against the immune-compromised host tissue (donor T-cells recognize the recipient's tissue as a foreign body [9]).Acute graft versus host disease is caused by an immune reaction of the donor cells to the host tissues and occurs within 100 days of the procedure and can be seen in 20-75% of allogenic BMT patients [9,13]. Because Acute GVHD is the result of human leukocyte antigen disparities, it does not occur with autologous BMT [13]. The most commonly affected organs are the skin, liver, and gastrointestinal tract [9,13]. Pulmonary involvement is rare [9,13]. The findings are non-specific with diffuse interstitial and alveolar opacities that resemble pulmonary edema [13]. Pulmonary hemorrhage can occur.
Chronic graft versus host disease occurs
beyond 100 days of the procedure and it is seen in 40-60% of
allogenic
marrow recipients who survive 6 months or more following BMT. It
is the most common complication in long-term survivors of BMT and
it occurs exclusively in allogeneic recipients as the result of an
immune-mediated reaction of the donor T-lymphocytes against host
tissues [13]. T-cell
depletion
of the donor marrow prior to infusion decreases the risk of GVHD
to
some
degree. GVHD is the most important factor predisposing BMT
patients to
infection, particularly with encapsulated bacteria. Unlike acute
GVHD, chornic GVHD commonly involves the lungs and can manifest as
organizing pneumonia or bronchiolitis obliterans [13]. Chronic
GVHD is
characterized
by thickening of the bronchovascular interstitium and patchy
ground
glass
opacifications. The incidence of late onset noninfectious
pulmonary
complications in patients with chronic GVHD is between 10-26%
[12].
Bronchiolitis obliterans is a late complication seen in
up to 10%(2% - 48%) of patients with chronic GVD typically 6 to 12
months
following
the procedure [13]. Patients present with progressive dyspnea, dry
cough, and
expiratory wheeze [9]. Pulmonary function tests
demonstrate a progressive, non-reversible airflow obstruction with
a FEV1
of less than 80% of predicted or a decrease in FEV1-forced
vital
capacity by 10% or more in under 1 year [9]. Bronchiolitis
obliterans syndrome describes a clinical syndrome characterized by
an irreversible decline in FEV1 of at least 20% from baseline-
these patients may not necessarily demonstrate classic pathologic
features of BO [13]. Mortality
from BO is high -up to 40% of affected patients secondary to
respiratory
failure. Other authors quote the mortality as between 12-27% at 5
years with most patients dying from a superimposed pulmonary
infection [13].Treatment includes steroids and immunosuppressant
therapies, but the overall response is poor [13]. Radiographically
BO is characterized by air-trapping which
produces a pattern
of mosaic attenuation, air trapping, and bronchial dilatation on
HRCT. The mosaic pattern is very suggestive of BO with a 74-91%
sensitivity and 67-94% specificity [13]. Small
(under 5 mm), ill-defined centrilobular nodules may also be
identified.
Non-classifiable (non-specific) Interstitial Pneumonia:
Non-classifiable (non-specific) interstitial pneumonia is
another,
less common manifestation of chronic GVHD [12]. Patients present
with progessive dyspnea and cough with a restrictive pattern
(reduced diffusion capacity) on PFTs [13]. The condition initially
manifests as areas of ground glass opaicty, particularly in a
peribronchial distribution, and then progresses to interstitial
changes, traction bronchiectasis, and honeycombing [12].
Acute Radiation Pneumonitis:
Acute radiation pneumonitis can manifest 6 weeks to 6 months
after radiation therapy [13]. Patients present with fever,
leukocytosis, and parenchymal infiltrates [13]. A distinctive
feature of the infiltrates is that they typically have a
well-demarcated border with normal lung that conforms to the
radiation port [13].
Pulmonary Cytolytic Thrombi:
Pulmonary cytolytic thrombi is a rare pulmonary complication that
exclusively affects allogeneic BMT patients with acute or chronic
GVHD [13]. The condition have been reported primarily in children
with a median onset of 72 days after BMT [13]. The condition
consists of intravascular thromboemboli associated with the
infiltration of monocytes of donor and recipient origins in small
and medium pulmonary vessels, surrounded by an area of alveolar
hemorrhage or infarct [13]. The thromboemboli are composed of
necrotic basophilic debris, with an amorphous material suggestive
of the products of cellular breakdown [13]. Symptoms are
nonspecific and include fever, dyspnea, and chest pain [13].
Patients respond well to an increase in immunosuppressive therapy,
with radiologic resolution over weeks to months 13].HRCT findings
consist of multiple, bilateral pulmonary nodules and peripheral
opacities that reflect lung infacrts [13].
Air Leak Syndrome:
Air leak syndrome (ALS) is a rare complication (1-6% of patients)
that refers to spontaneous pneumothorax, pneumomediastinum,
pulmonary interstitial emphysema, or subcutaneous emphysema in a
BMT recipient [13]. ALS typically occurs as a late complication of
allogeneic transplants in patients with chronic GVHD and
bronchiolitis obliterans syndrome [13]. ALS patients have an
overall poor prognosis which may be due to underlying BO [13].
Post-transplant Lymphoproliferative Disorder (PTLPD):
PTLD occurs in about 1-2% of BMT patients [9,13]. PTLD typically occurs within 6 months of BMT, but may occur as early as 30 days after transplant [13]. The disorder is a direct sequella of chronic immunosuppression with unchecked proliferation of Ebstein-Barr virus infected B-cells. The condition represents a spectrum of lymphoproliferative abnormalities ranging from polyclonal lymphoid hyperplasia to monoclonal B-cell lymphoma (typically a non-Hodgkin lymphoma [13]c xz). Affected patients nearly always have positive EBV-titers. The occurance of PTLD is directly related to immunosuppression- therefore, allogeneic recipients have an inherently higher risk for developing PTLD compared to autologous BMT recipients [13].PTLD most commonly involves the lymph nodes, spleen, and liver [13]. Pulmonary involvement occurs in about 20% of patients [13]. Treatment is reduction or elimination of immunosuppresive drugs, but mortality remains high in BMT patients. Radiologically the most common finding is the presence of multiple, well circumscribed pulmonary nodules (with or without mediastinal and hilar adenopathy) in association with enlarged tonsils or cervical adenopathy. Solitary nodules, and areas of interstitial or alveolar infiltrate have also been described.
Pulmonary Venoocclusive Disease (PVD):
Pulmonary venoocclusive disease is a rare complication of both autologous and allogeneic BMT [13]. PVD may be the result of endothelial injury caused by infection or drug toxicity [13]. The condition typically manifests 2-6 months following transplant with progressive dyspnea, fatigue, and pulmonary hypertension [13]. The triad of pulmonary hypertension, imaging findings of pulmonary edema, and normal pulmonary arterial occlusion pressure is strongly suggestive of the diagnosis [13]. The prognosis is poor and no specific treatment is available [13]. Imaging findings include dilatation of the main pulmonary artery, centrilobular ground-glass opaities, and smooth interlobular septal thickening 13]. Mediastinal lymph nodes are commonly enlarged [13].Pleuroparenchymal Fibroelastosis:
Pleuroparenchymal fibroelastosis is an extremely rare entity in allogeneic recipients characterized by thickenig of the pleura and fibroelastosis of the subpleural lung parenchyma [13]. The condition occurs 2-16 months after transplantation and patients present with recurrent pneumothorax [13].REFERENCES:
(1) Radiol Clin North Am 1991; 29(5): 983-997
(2) Radiol Clin North Am 1996; Jan 34(1): 97-117
(3) RadioGraphics 1997; Worthy SA, et al. Pulmonary complications after bone marrow transplantation: High-resolution CT and pathologic findings. 17: 1359-1371
(4) J Thorac Imag 1999; Conces DJ. Noninfectious lung disease in immunocompromised patients. 14: 9-24
(5) J Thorac Imaging 1999; McGuinness G. Viral and pneumocystis infections of the lung in the immunocompromised host. 14: 25-36
(6) J Thorac Imaging 1999; Connolly JE, et al. Opportunistic fungal pneumonia. 14: 51-62
(7) Radiology 1999; Leung AN, et al. Pulmonary infections after bone marrow transplantation: Clinical and radiographic findings. 210: 699-710
(8) J Thorac Imaging 1999; Choi YH, et al. Radiologic findings: Pulmonary infections after bone marrow transplantation. 14: 201-206
(9) AJR 2005; Freanquet T, et al. High-resolution CT and pathologic findings of noninfectious pulmonary complications after hematopoietic stem cell transplantation. 184: 629-637
(10) AJR 2005; Escuissato DL, et al. Pulmonary infections after bone marrow transplantation: high-resolution CT findings in 111 patients. 185: 608-615
(11) AJR 2006; Franquet T, et al. Thin-section CT findings in
hematopoietic stem cell transplant recipients with respiratory
virus
pneumonia. 187: 1085-1090
(12) AJR 2012; Song I, et al. CT findings of late-onset
noninfectious pulmonary complications in patients with
pathologically
proven graft-versus host disease after allogenic stem cell
transplant.
199: 581-587
(13) Radiographics 2014; Pena E, et al. Noninfectious pulmonary
complications after hematopoietic stem cell transplantation:
practical approach to imaging diagnosis. 34: 663-683