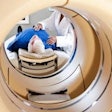
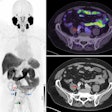
The Food and Drug Administration has given 510(k) clearance to GE Medical Systems of Waukesha, WI, for the mobile version of its Discovery ST PET/CT system. The fixed-site version of the scanner was cleared last October.
Discovery ST has a larger bore than GE's flagship Discovery LS system. Discovery ST supports both 2-D and 3-D imaging and is available in either a four-slice or eight-slice CT configuration. Shipments are expected to begin in the second quarter of this year, at a list price of between $1.8 million to $2.1 million, depending on configuration. The mobile version will be priced at $100,000 more, plus cost of the transport van.
GE's first hybrid PET/CT system, Discovery LS, which has 16-slice capability, will remain in the product line. The company just received a second 510(k) for this system that allows GE to market the scanner for the diagnosis of cancer, and specifically for neurology and cardiology. Previously, such statements could not be made by the OEM, but only by an interpreting physician.
By AuntMinnie.com staff writers
March 31, 2003
Related Reading
German radiologists assess performance of PET-CT, March 10, 2003
GE touts advances in PET/CT instrumentation, December 3, 2002
In-line PET-CT system improves cancer diagnosis, October 10, 2002
Copyright © 2003 AuntMinnie.com