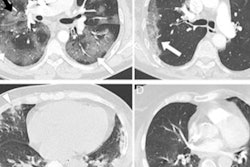
The U.S. Food and Drug Administration (FDA) is curtailing inspection activities for the safety of employees in response to the continuing spread of the COVID-19 omicron variant.
Through January 19, the agency intends to continue mission-critical international and domestic inspections, but it has temporarily postponed certain inspectional activities with the hopes of restarting these activities as soon as possible. State inspections under FDA contracts have the discretion to make inspection decisions based on their local information, the agency said.
In addition, the agency also is postponing the planning of prioritized surveillance of overseas inspection assignments that were scheduled to begin in February 2022.