CAPE TOWN -- Diffusion weighted imaging (DWI) with diffusion kurtosis can help monitor early response prediction and personalized therapy in breast cancer patients, suggest findings presented at the International Society for Magnetic Resonance in Medicine (ISMRM) meeting.
In her May 13 presentation, Debosmita Biswas from the University of Washington in Seattle shared her team’s findings on how DWI markers can accurately predict early pathologic response in breast tumors treated with neoadjuvant chemotherapy compared to conventional methods.
“The eventual goal would be for us to utilize these DWI quantitative metrics for individualized breast cancer treatment planning,” Biswas told AuntMinnie.
Early prediction of breast cancer response to therapy is needed to guide treatment decisions. The conventional REsponse Criteria In Solid Tumors (RECIST) method relies on tumor size changes over time. However, Biswas noted that this method is not sensitive to early changes in tumor microstructure before shrinkage becomes visible.
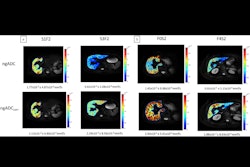
DWI has shown promise in this area by providing more specific insights into tumor microenvironments. Biswas and colleagues studied predictive values of multiple DWI models to identify imaging biomarkers for early prediction of pathologic complete response to neoadjuvant chemotherapy.
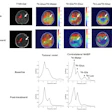
The multisite study included 37 women who completed MRI exams, chemotherapy, and surgery. Multiparametric 3T MRI scans were performed at four timepoints: baseline, early-treatment (after one treatment cycle), mid-treatment, and post-treatment (pre-surgery).
Most cancers consisted of invasive ductal carcinoma (92%) and high-grade cancers (73%), and 15 women achieved pathologic complete response.
Between baseline and early treatment scans, tumor longest diameter decreased by 4 mm, while percentage change in longest diameter showed “fair” predictive performance for pathologic versus non-pathologic complete response (area under the curve [AUC] = 0.7; p = 0.04).
Biswas also reported the following:
Among baseline features, only C1 and C2 from restriction spectrum imaging (RSI) showed marginally significant predictive value for pathologic complete response (AUC=0.73 and 0.68; p = 0.053 and 0.046, respectively).
Across the DWI models, percentage change in diffusivity measures were highly predictive of complete response [Dk (AUC = 0.94; p = 0.003), apparent diffusion coefficient (AUC = 0.89; p = 0.01), Dt (AUC = 0.85, p = 0.01)].
Percentage change for C1 (AUC = 0.8; p = 0.01) and impeded diffusion fraction (AUC = 0.7; p = 0.01) also exceeded the predictive performance of tumor longest diameter.
Percentage changes in other markers did not achieve statistical significance.
The researchers also used leave-one-out cross-validation to estimate how well the models performed on unseen data. Through this, Biswas reported that the DWI model incorporating percentage change on diffusion kurtosis achieved an AUC of 0.91 with minimal optimism (3.5%). This points to “robust” performance with low risk of overfitting.
Finally, analysis on Least Absolute Shrinkage and Selection Operator (LASSO) regression identified percentage change in diffusion kurtosis as the best predictive feature (AUC = 0.94) with high sensitivity (93.3%) and specificity (90.9%).
Biswas said the results support incorporating advanced DWI metrics into breast cancer treatment monitoring. She also called for larger multi-institutional cohorts to validate these findings for clinical translation.
“We need to standardize our MR DWI protocols and I think it just needs to be done across, not just in specialized institutions like ours, but across community hospitals,” Biswas told AuntMinnie. “All of those people need to be familiar and confident about using their DWI protocols. I think that would be a great leap toward clinical care.”
The study earned Summa Cum Laude honors from the ISMRM Annual Meeting Program Committee.
Check out AuntMinnie’s full coverage of ISMRM 2026 on our ShowCast.