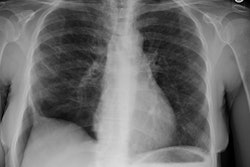
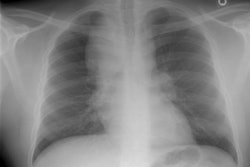
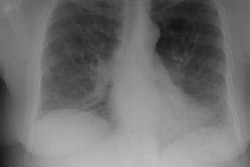
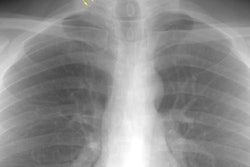
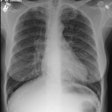
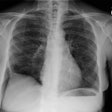
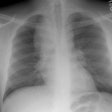
Interstitial Pneumonitis/ Idiopathic Pulmonary Fibrosis/ Fibrosing Alveolitis:
Incidentally detectable lung abnormalities (ILAs) refers to a
sublte or mild parenchymal abnormality identified on CT in up to
9% of patients over the age of 50 years in whom interstitial lung
disease was not previously clinically suspected [44]. Subjects
with ILA tend to be older, have had increased exposure to tobacco
smoke, and are twice as likely to report having a chronic cough
and shortness of breath [44]. Patients with ILA have been shown to
have a 12% decrease in diffusion capacity for carbon monoxide and
a 9% decrease in total lung capacity compared to patients without
ILA [44]. ILA has been shown to progress over time [44].
The clinical term idiopathic pulmonary fibrosis (or cryptogenic fibrosing alveolitis) describes a progressive disorder of the lung parenchyma characterized by fibrosis and variable intra-alveolar and interstitial exudate. It is often used interchangeably with the histologic terms desquamative interstitial pneumonitis (DIP) and usual interstitial pneumonitis (UIP). However, once a histologic diagnosis has been obtained, it is more appropriate to use the histologic terminology. The distinction between DIP and UIP is controversial- some authors feel that DIP is an early manifestation of UIP, while others feel that it is a distinct clinical entity. In patients with interstitial pneumonitis, determinants for a favorable prognosis include a younger age at onset, female sex, a normal oxygen saturation, a near normal chest radiograph, a cellular alveolar infiltrate on histologic analysis (DIP), and underlying progressive systemic sclerosis (scleroderma).
Other disorders considered in this category of interstitial pneumonias include acute interstitial pneumonia (AIP- previously referred to Hamman-Rich syndrome), non-specific interstitial pneumonia (NIP), respiratory bronchiolitis with interstitial lung disease (RB-ILD) - which some authors feel is part of the spectrum of DIP, lymphoid interstitial pneumonia (LIP- which some consider a lymphoproliferative disorder), and cryptogenic organizing pneumonia (COP- also referred to as BOOP) [17].
Desquamative Interstitial Pneumonia:
Alveolar macrophage pneumonia
View Cases of DIP
Clinical:
Desquamative interstitial pneumonitis (DIP) may be a self limited disease or may progress to usual interstitial pneumonitis (UIP), although progression to UIP is controversial [8]. Affected patients are typically 8 to 10 years younger than those with UIP (age 30-40 years[24]) and about 90% of affected patients are smokers. Currently, DIP is felt to be a direct result of smoking-related lung toxicity [18]. Other smoking related lung diseases in this spectrum include respiratory bronchiolitis and respiratory bronchiolitis-interstitial lung disease [19]. The disorders differ histologically in that DIP is diffuse, while respiratory bronchiolitis and RB-ILD are centered on the respiratory bronchiole [19]. Some authors feel that DIP may represent the end spectrum of RB-ILD [26].
DIP is much less common than UIP. Men are affected twice as frequently as women [19,26]. Although patients may be asymptomatic, they can present with slowly progressive dyspnea, non-productive dry cough, or digital clubbing (in up to 40% of cases) [19]. Pulmonary function testing reveals a restrictive abnormality and decreased diffusing capacity (DLCO) - with reductions of 50% or more being common [17,26]. Bronchoalveolar lavage fluid contains increased numbers of alveolar macrophages with granules of "smoker's pigment" [17]. Treatment consists of smoking cessation and steroids [18]. Response to therapy is seen in nearly 60% of cases- which is better than UIP. Overall, patients with DIP have a much better prognosis than those with UIP- the five year mortality is about 5% (and an overall survival of 70% at 10 years [19]). Late relapse and recurrence in a transplanted lung have been reported [26]. Unfortunately, only about 15% of patients with interstitial pneumonitis present with histologic findings suggestive of DIP.
DIP can be distinguished from UIP on histologic analysis which reveals an abundance of pigmented macrophages within the air spaces, minor fibrotic changes, and preservation of the alveolar architecture [26]. As opposed to the bronchocentric distribution in RB-ILD, lung involvement in DIP is more diffuse and uniform [24]. DIP is considered a misnomer as the predominant pathologic feature is intraalveolar accumulation of pigmented macrophages and not desquamation of epithelial cells as previously thought [26].
X-ray:
CXR: On CXR there are usually linear irregular opacities predominantly in the bases (60%) with preserved lung volumes, but the film can be normal (15-20%). A basal ground glass alveolar pattern sparing the costophrenic angles is seen in 20%. Diffuse ground glass opacities occur in 15%. The CXR can be normal in 3 to 22% of cases.
Computed tomography: On HRCT areas of patchy ground glass attenuation within the lungs are the predominant finding and is present in all cases [8,11] (reflecting the presence of intra-alveolar macrophages and interstitial inflammation). The opacities are typically bilateral, symmetric (up to 85% of cases), peripheral/subpleural (59% of cases), and predominantly involve the lung bases/low lung zones in a majority of cases (73%) [38]. The appearance can be indistinguishable from that seen in acute hypersensitivity pneumonitis. Other disorders which produce a similar appearance include respiratory bronchiolitis interstitial lung disease, drug toxicity, BOOP, and pulmonary alveolar proteinosis. The ground-glass opacities usually regress with therapy [19].
If present, findings of fibrosis (intra/interlobular septal thickening) are limited in extent and relatively mild. However, other authors suggest that a limited reticular pattern and some irregular linear opacities are frequent (59% of cases) [38]. Small cysts may be seen in the areas of ground glass opacification and represent dilated bronchioles, reversible bronchiolectasis, and dilated alveolar ducts [8,38].
Usual Interstitial Pneumonia:
View cases of UIP
Clinical:
UIP is the most common form of chronic interstitial pneumonitis
(10 to 20 times more prevalent than DIP). Most cases occur
sporadically, although familial forms have been reported. Patients
typically present between the ages of 50-70 years with progressive
dyspnea and a dry cough [19]. Clinical findings include late
inspiratory velcro rales, clubbing (50-80%), restrictive defects
on pulmonary function testing, a decreased diffusion capacity for
carbon monoxide, decreased PAO2 with rest or
exercise, and lymphocytosis on lavage. Increased BAL eosinophils
can be seen and suggest a poor prognosis [6]. Men are affected
slightly more frequently than women [21].
Etiologies of UIP:
Idiopathic: UIP may be idiopathic (50% of cases)
Familial (earlier age of onset, mode of inheritance unknown) [12]
Drug exposure (bleomycin, cyclophosphamide, methotrexate, Adriamycin).
Collagen vascular disease: Between 20-30% of patients with UIP
have an underlying collagen vascular disease (rheumatoid arthritis
[most commonly], systemic sclerosis, or
dermatomyositis/polymyositis). Involvement of the anterior upper
lobes is suggestive of CVD-associated UIP [43].
Smoking: A history of cigarette smoking seems to be a risk factor for the development of IPF and the risk increases with duration of smoking [24,26,36]. About 60% of patients with IPF are current or former smokers [39]. Smoking, however, does not appear to affect the course of the disease [24]- although, more recent studies suggest decreased survival in smokers with IPF [26]. Changes of pulmonary fibrosis can be found in up to 10% of smokers that undergo lung cancer screening [36].
GERD: Asymmetric IPF can be associated with GERD and the disease
is typically worse on the patient's preferred sleeping side [42].
Patients with asymmetric disease have twice the incidence of
reflux compared to patients with symmetric disease [42].
A limited predominantly subpleural reticular pattern can be seen in a large percentage (up to 60%) of asymptomatic older patients (over 75 years) and this may represent the normal spectrum of morphology for the aging lung [31].
The histologic diagnosis of UIP is based upon temporal heterogeneity: the identification of fibrotic lesions at different stages [19].
UIP treatment: For patients with idiopathic pulmonary fibrosis
response to steroid therapy is rare [13]. Steroid therapy may
result in improvement in the appearance of ground glass opacities,
but progression to end-stage lung disease does not seem to be
affected by therapy [1,7]. In fact, the host of side effects
associated with high-dose corticosteroid therapy may cause more
harm than good in many of these patients [13]. Treatment with
cytotoxic agents have been shown to be of only limited benefit. A
potential new therapy is interferon-γ-1b
which inhibits fibroblast proliferation and collagen synthesis
[17]. Therapy with n-acetylcysteine has been shown to be
associated with decreased lung function deterioration over 1 year
(the agent inhibits adenosine deaminase enzyme and restores a more
normal oxidant-antioxidant balance) [32]. Two medications,
pirfenidone (Esbriet, InterMune) and nintedanib (Ofev, Boehringer
Ingelheim), have been approved for the treatment of IPF in the
United States [41].
Lung transplantation is a therapeutic alternative in selected patients. Prognosis is poor, with a five year survival of approximately 40-50% following the onset of dyspnea (the median survival after diagnosis is 2 to 3.5 years [15,19]). For patients with idiopathic pulmonary fibrosis, those with fibrosis involving more than 25% of the lungs (as determined by HRCT) have the worst prognosis [13]. Some patients have phases of accelerated deterioration superimposed upon the chronic course of their illness.
Complications of UIP:
1- Lung cancer: Patients with UIP are at an increased risk for
bronchogenic carcinoma [34]. Between 10-15% of patients may
develop bronchogenic carcinoma [19]- most commonly adenocarcinoma.
The tumors tend to occur in areas of fibrosis at the interface
between the normal and fibrotic lung [34]. Most cancers are
located at the interface between fibrotic and normal lung or in
the midst of fibrotic cysts of the lower subpleural lungs [37].
The tumors are often stellate or bandlike in shape and can be
difficult to identify due to the underlying fibrosis [34].
2- Accelerated deterioration or exacerbation is another potential
complication (with a decrease in arterial oxygen tension (PaO2)
of more than 10 mm Hg in one month [19,21]. Accelerated
deterioration manifests with a rapid onset of dyspnea in a patient
with underlying IPF [19]. There is usually a short prodome of 4-8
weeks duration [19]. On CT, there is diffuse or peripheral
ground-glass opacification superimposed upon the patients
underlying interstitial lung disease [19].
3- Pulmonary arterial hypertension can occur in up to 46% of patients and is associated with a worse prognosis [27]. In patients with pulmonary fibrosis, pulmonary artery dilatation can occur in the absence of PAH and therefore, an enlarged PA cannot be used to reliably confirm or exclude the diagnosis of PAH [27].
Combined pulmonary fibrosis and emphysema is being increasing
recognized as a distinct clinical entity in smokers [26]. Patients
are almost exclusively men in their 6th and 7th decades [26]. Lung
volumes are relatively preserved despite markedly impaired
diffusion capacity and hypoxemia during exercise [26].
Honey-combing, reticular opacities, and traction bronchiectasis
affecting the lower lung zones, with upper lobe paraseptal and
centrilobular emphysema are the most frequent findings on HRCT
[26].
Interstitial lung abnormalities (ILAs) are defined as
non-dependent interstitial changes in asymptomatic patients
affecting more than 5% of any lung zone and can be seen in older
individuals (4-9% of smokers and 2-7% of non-smokers) [45]. Other
authors suggest that subpleural basal reticular changes can be
found in up to 60% of patients older than 75 years [45].
X-ray:
CXR: A ground glass pattern may be seen on CXR in the early stages of the alveolitis and may reflect the more cellular form of the disorder [DIP]. Later a there is a reticular pattern/ reticulonodularity predominantly in the bases with a shaggy heart boarder and loss of lung volume. Honey comb lung represents end-stage disease. Up to 10% of patients with diffuse interstitial lung disease can have normal chest radiographs and CT is much more sensitive (88-95% compared to about 80% by CXR) in the detection of interstitial lung disease. Nonetheless, a normal HRCT does not exclude the diagnosis.
HRCT: UIP produces very characteristic findings on HRCT
which can permit the proper diagnosis without the requirement for
a lung biopsy [19]. The current guidelines support three
categories of UIP diagnosis on chest CT: definite UIP, possible
UIP, and inconsistent with UIP [39]. The American Thoracic Society
and the European Respiratory Society concluded that the presence
of characteristic features of IPF or UIP at thin-section CT allows
a confident diagnosis and precludes the need for surgical biopsy
[23]. To establish a definite UIP diagnosis there must be
reticular pulmonary fibrosis that is basilar and sub-pleural
predominant, honeycombing with or without traction bronchiectasis,
and no other features that suggest another diagnosis (such as
cysts, nodules, substantial air trapping, or predominant ground
glass attenuation) [39,40]. Possible UIP has all the features of
definite UIP, but there is no honeycombing [39].
HRCT criteria for UIP pattern [42]:
A. Definite UIP:
- Subpleural, basal predominant
- Reticular abnormality
- Honeycombing with or without traction bronchiectasis
- Absence of findings inconsistent with UIP listed in C below
B. Possible UIP
- The findings in A, but with the exception of honeycombing
C. Inconsistent with UIP (any of the seven features)
- Upper or midlung predominance
- Preibronchovascular predominance
- Extensive ground-glass abnormality (greater than the extent of
reticulation)
- Profuse micronodules (bilateral, predominantly upper lobes)
- Discrete cysts (multiple, bilateral, away from areas of
honeycombing)
- Diffuse mosaic attenuation/air-trapping (bilateral, in three or
more lobes)
- Consolidation in bronchopulmonary segment(s)/lobe(s)
In about 50% of cases, HRCT scan is sufficient to allow a confident diagnosis of IPF and lung biopsy can be avoided [21]. The sensitivity of CT for the diagnosis of IPF is about 63% and the specificity is about 70% [21]. The positive predictive value of a HRCT diagnosis of UIP is between 70-100% [19]. The sensitivity, specificity, and accuracy for the diagnosis of UIP versus NSIP is decreased in patients with underlying emphysematous changes [29]. Emphysematous spaces surrounded by ground-glass opacities can mimic honey-comb cysts due to accentuation of the interstitial spaces between areas of emphysema [29].
The interstitial changes of UIP are predominantly peripheral. A hallmark of UIP is it's patchy distribution with a predilection for the lung bases (an apicobasal gradient [24])- particularly the sub-pleural regions of the posterobasal segments of the lower lobes (the central lung zones are spared until late in the course of the disorder). Intralobular interstitial thickening is most commonly seen in patients with idiopathic pulmonary fibrosis and it produces a reticular or web-like pattern characteristically in the sub-pleural lung periphery. Associated irregular septal thickening and intralobular (centrilobular) bronchovascular thickening are sometimes noted. Irregular perivascular interstitial thickening is common and produces irregular interfaces between the lung and the pulmonary vessels. Subpleural lines can be seen, and may resolve following initiation of therapy- possibly representing resolution of atelectatic changes due to decreased lung compliance.
In the setting of interstitial lung disease the presence of areas of ground glass opacity have been shown to reflect active parenchymal inflammation (in 65-85% of cases), provided that these areas are not associated with evidence of fibrosis, traction bronchiectasis, or bronchiolectasis. This is because ground glass attenuation may merely reflect chronic interstitial fibrosis which appears as ground glass density due to partial volume averaging with cystic changes that may be too small to be resolved anatomically [11].
Late in the disorder there is honeycombing, traction bronchiectasis, and considerable architectural distortion. Honeycombing can be found in up to 96% of patients with end-stage idiopathic pulmonary fibrosis. The honeycomb cysts usually range from 2 to 20 mm, typically share walls, and frequently occur in several layers in the sub-pleural lung. A normal HRCT does not exclude early interstitial lung disease and can be seen in up to 12% of patients [5]. Pleural disease is not a feature of UIP. Occasionally, dense subpleural pulmonary fibrosis may mimic pleural thickening.
The presence of enlarged mediastinal lymph nodes was once classically felt to be seen in association with scleroderma. However, enlarged mediastinal nodes can be found in 55% to 66% of patients with idiopathic pulmonary fibrosis [22]. Other authors indicate that mild mediastinal lymph node enlargement can be seen in approximately 70% of patients with IPF [38]. Patients on steroid therapy have a much lower incidence of mediastinal adenopathy [22].
PET: Increased pulmonary FDG uptake has been described in patients with idiopathic pulmonary fibrosis and is even identified in areas of reticulation/honeycombing [30].
Nonspecific Interstitial Pneumonia:
View cases of NIP
Clinical:
NIP describes a group of interstitial pneumonias which cannot be
classified histologically as UIP, DIP, AIP, or BOOP. NSIP is the
second most common cause of idiopathic interstitial pneumonia-
accounting for 14-36% of cases [28]. The main features that
separate this disorder from the more usual interstitial pneumonia
(UIP) are the more temporal homogeneity of the histologic
process (occurring over a narrow time span) and a more benign
clinical course with a substantially better prognosis [15,19]. The
disorder is differentiated from DIP by the lack of significant
intraalveolar macrophage accumulation [9]. Affected patients may
have associated collagen vascular disease (16%) or history of
noxious agent exposure (17%). NSIP is the most common histologic
pattern of interstitial lung disease in patients with a collagen
vascular disorder (systemic sclerosis, RA, Sjogrens syndrome)
[28,33,35]. Several drugs (amiodarone, bleomycin, methotrexate,
and carmustine) can also produce NSIP [20]. Patients are most
commonly 40-50 years of age- about a decade younger than UIP
patients [24]- and present with dyspnea, dry cough, and low grade
fever. There is no gender predilection and cigarette smoking is
not an obvious risk factor [24,28]. The average duration of
illness is about 8 months. Bronchoalveolar lavage fluid reveals an
increase in the percentage of lymphocytes [17].
Pathologically NSIP is characterized by spatial and temporal
uniform interstitial inflammation with varying degrees of fibrosis
[28]. NIP is subdivided into two sub-categories: 1- cellular (less
common, but better prognosis and response to corticosteroids) and
2- fibrotic (generally worse prognosis with this subtype)
[11,19,24]. Treatment is based on the use of steroids and
cytotoxic drugs such as cyclophosphamide and cyclosporin [24].
Prognosis is significantly better than for patients with UIP- with about 50% of patients responding to steroid therapy. [2,3] The 5 year survival rate for cellular NSIP is nearly 100%; while the survival for fibrotic NSIP is 45-90% at 5 years, and 35% at 10 years (which is still better than for UIP) [28]. Following treatment, the abnormalities seen on CT (including reticular abnormalities and traction bronchiectasis) completely or partially resolve [15]. Up to half of NSIP cases also include areas of organizing pneumonia, however, in NSIP the areas of organizing pneumonia should involve less than 20% of the biopsy specimen [28].
X-ray:
CXR: Irregular linear opacities and air-space consolidations have been described in association with NIP.
Computed tomography: NSIP has a variable appearance at HRCT [14]. The lower lobes are more frequently involved (84-95% of patients [28,38]) and an obvious apicobasal gradient (as seen in UIP) is usually missing [24]. Diffuse involvement can be seen in 6-16% of cases [28]. The disorder can be peripheral (38-74% of patients) or demonstrate an axial, peribronchovascular distribution with a characteristic subpleural sparing in 21-64% of cases [28,33,38,42]. One of the findings that can aid in the differentiation of NSIP from UIP is the presence of "subpleural sparing" (found in up to 43% of cases) [25]. Bilateral, relatively symmetric disease is another finding associated with NSIP [28]. The interstitial fibrosis is usually fine and produces a thin reticular appearance [28]. Reticular abnormalities are found in 80-94% of patients [28].
Bilateral, patchy ground glass opacities are found in 76% to 100% of cases [15]. Areas of ground-glass attenuation are symmetric in up to 86% of patients and are commonly subpleural (68%) [14]. The presence of ground-glass abnormalities aids in distinguishing NSIP from UIP- ground-glass abnormalities involving more than 15% of the lung is felt to be a significant predictor for the presence of NSIP rather than UIP [29]. The ground-glass attenuation is frequently associated with bronchiectasis [15]. Predominantly basilar, subpleural air space consolidation is also very common (16-70% of cases)- although other authors indicate that consolidation is uncommonly the primary abnormality in NSIP [28].
Irregular linear opacities are found in 30-87% of cases and are subpleural in up to 96% [14] of cases. Mediastinal adenopathy can be found in 4-30% (up to 81% [22]) of cases. Honeycombing is generally not a feature of this disorder [9,19,25], although it was found in 26% to 30% of cases in some studies [14,16]. Small (under 5 mm), bilateral, poorly defined, centrilobular nodules may be seen in 14% of cases [14], but are more common in other disorders such as RB-ILD or hypersensitivity pneumonitis [28]. On following up exams after treatment, areas of ground-glass opacification will decreased or resolve [10]. However, up to 28% of patients demonstrate progressive abnormalities suggestive of IPF [25].
Overall, HRCT accuracy for the diagnosis of NSIP is variable - with a sensitivity of 70%, a specificity of 63%, and a correct diagnosis in 41-86% of patients [28,38]. However, there is also high interobserver variability among pathologists for the diagnosis of NSIP [28].
REFERENCES:
(1) Radiology 1993 Dec;189(3):687-691
(2) RadioGraphics 1996;16:1009-1033
(3) J Thorac Imag 1996; 11: 231-249
(6) J Comput Assist Tomogr 1997; Kim Y, et al. The spectrum of eosinophilic lung disease: Radiologic findings. 21 (6): 920-930
(7) J Thorac Imaging 1998; Lee JS, et al. Usual interstitial pneumonia: Relationship between disease activity and the progression of honeycombing at thin-section computed tomography. 13: 199-203
(8) Thorax 1997; Akira M, et al. Serial computed tomographic evaluation in desquamative interstitial pneumonia. 52: 333-337
(9) AJR 1998; Kim TS, et al. Nonspecific interstitial pneumonia with fibrosis: High-resolution CT and pathologic findings. 171: 1645-1650
(10) AJR 1999; Kim EY, et al. Non-specific interstitial pneumonia with fibrosis: Serial high-resolution CT findings with functional correlation. 173: 949-953
(11) Society of Thoracic Radiology Course Syllabus 2000; Lynch DA, et al. International concensus classification of idiopathic intestitial pneumonias. 52-56
(12) Thorax 2000; Marshall RP, et al. Adult familial cryptogenic fibrosing alveolitis in the United Kingdom. 55: 143-146
(13) Chest 2000; Michaelson JE, et al. Idiopathic pulmonary fibrosis. A practical approach for diagnosis and management. 118: 788-794 (No abstract available)
(14) Radiology 2000; Hartman TE, et al. Nonspecific interstitial pneumonia: Variable appearance at high-resolution chest CT. 217: 701-705
(15) Radiology 2001; Lynch DA. Nonspecific interstitial pneumonia: evolving concepts. 221: 583-584 (No abstract available)
(16) Radiology 2002; Johkoh T, et al. Nonspecific interstitial pneumonia: correlation between thin-section CT findings and pathologic subgroups in 55 patients. 225: 199-204
(17) Radiographics 2003; Wittram C, et al. CT-histologic correlation of the ATS/ERS 2002 classification of idiopathic interstitial pneumonias. 23: 1057-1071
(18) AJR 2005; Miller WT, Shah RM. Isolated diffuse ground-glass opacity in thoracic CT: causes and clinical presentations. 184: 613-622
(19) Radiology 2005; Lynch DA, et al. Idiopathic interstitial pneumonias: CT features. 236: 10-21
(20) Radiol Clin N Am 2005; Lindell RM, Hartman TE. Chest imaging in iatrogenic respiratory disease. 43: 601-610
(21) AJR 2005; Souza CA, et al. Idiopathic pulmonary fibrosis: specturm of high-resolution CT findings. 185: 1531-1539
(22) AJR 2006; Souza CA, et al. Idiopathic interstitial pneumonias: prevalence of mediastinal lymph node enlargement in 206 patients. 186: 995-999
(23) Radiology 2006; Sumikawa H, et al. Usual interstitial pneumonia and chronic idiopathic interstitial pneumonia: analysis of CT appearance in 92 patients. 241: 258-266
(24) Radiographics 2007; Mueller-Mang C, et al. What every radiologist should know about interstitial pneumonias. 27: 595-615
(25) Radiology 2008; Silva CI, et al. Nonspecific interstitial pneumonia and idiopathic pulmonary fibrosis: changes in pattern and distribution of disease over time. 247: 251-259
(26) Radiographics 2008; Attili AK, et al. Smoking-related interstitial lung disease: radiologic-clinical-pathologic correlation. 28: 1383-1398
(27) Radiology 2008; Devaraj A, et al. The effect of diffuse pulmonary fibrosis on the reliability of CT signs of pulmonary hypertension. 249: 1042-1049
(28) Radiographics 2009; Kligerman SJ, et al. Nonspecific interstitial pneumonia: radiologic, clinical, and pathologic considerations. 29: 73-87
(29) Radiology 2009; Akira M, et al. Usual interstitial pneumonia and nonspecific interstitial pneumonia with and without concurrent emphysema: thin-section CT findings. 251: 271-279
(30) J Nucl Med 2009; Groves AM, et al. Idiopathic pulmonary fibrosis and diffuse parenchymal lung disease: implications from initial experience with 18F-FDG PET/CT. 50: 538-545
(31) Radiology 2009; Copley SJ, et al. Lung morphology in the
elderly: comparitive CT study of subjects over 75 years old versus
those under 55 years old. 251: 566-573
(32) J Nucl Cardiol 2011; Grady EC, et al. Development of
asystole requiring cardiac resuscitation after the administration
of regadenoson in a patient with pulmonary fibrosis receiving
n-acetylcysteine. 18: 521-525
(33) Radiographics 2012; Capobianco J, et al. Thoracic
manifestations of collagen vascular diseases. 32: 33-50
(34) AJR 2012; Yoshida R, et al. Lung cancer in chronic
interstitial pneumonia: early manifestations from serial CT
observations. 199: 85-90
(35) AJR 2013; Nemec SF, et al. Lower lobe-predominant diseases
of the lung. 200: 712-728
(36) Radiology 2013; Jin GJ, et al. Interstitial lung
abnormalities in a CT lung cancer screening population: prevalence
and progression rate. 268: 563-571
(37) AJR 2015: Oh SY, et al. Evolving early lung cancers detected
during follow-up of idiopathic interstitial pneumonia: serial CT
features. 204: 1190-1196
(38) Radiographics 2015; Sverzellati N, et al. American thoracic
society-European respiratory society classification of the
idiopathic interstitial pneumonias: advances in knowledge since
2002. 35: 1849-1872
(39) AJR 2016; Chung JH, Lynch DA. The value of a
multidisciplinary approach to the diagnosis of usual interstitial
pneumonitis and idiopathic pulmonary fibrosis: radiology,
pathology, and clinical correlation. 206: 463-471
(40) AJR 2016; Gruden JF, et al. Diagnosis of usual interstitial
pneumonitis in the absence of honeycombing: evaluation of specific
CT criteria with clinical followup in 38 patients. 206: 472-480
(41) AJR 2016; Salatore M, et al. Evidence of interstitial lung
disease on low-dose chest CT images: prevalence, patterns, and
progression. 206: 487-494
(42) AJR 2016; Gruden JF. CT in idiopathic pulmonary fibrosis:
diagnosis and beyond. 206: 495-507
(43) AJR 2018; Chung JH, et al. CT features of the usual
interstitial pneumonia pattern: differentiating connective tissue
disease-associated interstitial lung disease from idiopathic
pulmonary fibrosis. 210: 307-313
(44) Radiology 2019; Hatabu H, et al. Interstitial lung
abnormality: recognition and perspectives. 291: 1-3
(45) Radiology 2019; Goo JM. Interstitial lung abnormalities:
poor prognosis for patients with lung cancer. 292: 499-500