Silicosis:
Clinical:
Silicosis results from the inhalation of crystalline silica (silicon dioxide) which can occur in occupations such as mining, quarrying, and tunneling [7]. The fibrogenic forms of silica are cristobalite and tridymite [7]. Silicosis has a tendency to progress even after exposure is stopped. The silica particles are ingested by pulmonary macrophages which cause the macrophage to break-down with release of it internal enzymes. The inflammatory response eventually leads to fibrogenic changes or formation of the "silicotic nodule", which has a characteristic basket weave appearance. Eventually, the particles are transported to regional nodes. There are 5 types of disease:
1- Simple Silicosis: Requires a 10 to15 year exposure. Most patients are asymptomatic when first seen, but may have a productive cough. Approximately 20 to 30% of cases will progress to complicated silicosis. Simple silicosis may be complicated by a superimposed TB infection due to macrophage dysfunction in these patients.
2- Accelerated silicosis: The appearance of accelerated silicosis is similar to simple disease, however, the clinical progression is more rapid- occurring within 4-10 years of heavier exposure [1,8].
3- Complicated Silicosis: Requires a period of 10 to15 years to progress from simple disease to complicated silicosis (progression can occur even in the absence of continued exposure). The disorder is characterized on radiographs by 'conglomerate masses' which represent a coalescence of smaller nodules. Intense FDG uptake can be seen in the conglomerate masses on PET imaging [7]. Death is usually the result of cardio-respiratory failure.
4- Silico-tuberculosis: There is a higher incidence of TB in patients with silicosis (up to 25% of patients [7]) most likely due to impaired macrophage function. Superimposed TB should be suspected if there is rapid progression of simple disease, asymmetric nodules or consolidation, or cavitation [7].
5- Acute Silicosis (Silico-proteinosis): Acute sikicosis is a fulminating respiratory illness resulting from relatively short exposure to high concentrations of fine silica dust [8]. In the disorder, massive silica dust inhalation overwhelms the lungs clearing mechanisms. The silica particles are directly toxic to pneumocytes of the alveolar walls and the alveoli are filled with a PAS positive staining proteinaceous material (similar in character to that found in alveolar proteinosis), blood, and debris. The intense exposure is generally over a 6 to 8 month period, but can follow a single event. The condition usually manifests within 3 years of the initial exposure with rapid progression of shortness of breath often associated with constitutional symptoms (dyspnea, dry cough, cyanosis, fever, and weight loss) [8]. The disorder most commonly affects sandblasters, but individuals in mining operations, stone cutting, stone polishing, foundry work, fly ash exposure, and glass making are also at risk for development of this disease. Sand is also used in the manufacture of paints, cosmetics, and plastics. Tissue obtained via transbronchial or open lung biopsy will demonstrate the silica particles. Silicotic nodules are smaller, less frequent, or may be completely absent. Superimposed mycobacterial infection can be a complication. The prognosis is reported to be poor and the disease is often rapidly progressive with death (often within 2 years[8]) secondary to respiratory failure. Transient clinical and radiographic improvement have been described in association with steroid therapy [3].
Radiographically there are large areas of alveolar parenchymal consolidation and/or ground-glass opacities especially in the upper lobes, which can mimic pulmonary edema. Air bronchograms can be seen within the opacities. If the patient survives, marked fibrosis may result. Hilar adenopathy is uncommon. The most common findings on CT is that of bilateral airspace consolidations mainly involving the posterior aspects of the lungs and often (75% of cases) associated with multifocal ground glass opacities [8]. Innumerable, small, ill-defined, centrilobular nodules (either soft-tissue or ground-glass attenuation) can be found in up to 85% of cases- reflecting the bronchocentric nature of the disorder [8]. The nodules can become confluent in a large percentage of patients and foci of calcification can be seen within the areas of consolidation in up to 83% of cases [8]. Sometimes a "crazy-paving" appearance can be seen [1,2,4,7] Calcified lymph nodes can be identified in up to 85% of patients [8].
X-ray:
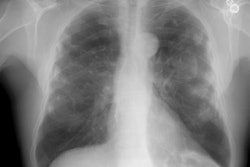
CXR: On plain film the characteristic radiographic finding is multiple, well-defined small nodules (2-5 mm in size) (they may appear sand-like). The disease has a tendency to involve the mid and upper lung zones- particularly the posterior portions [5]. These small nodules may calcify in 10-20% of cases [7]. A combination of small nodules and a reticular pattern can also be seen. Later, there is dense, nodular bilateral upper lobe fibrosis. Differential considerations for upper lobe fibrosis include: (C-A-S-S-E-T) C- Cystic fibrosis; A- Aspergillosis; S- Sarcoid; S- Silicosis; E- Eosinophilic granuloma; and T- TB.
A conglomerate mass (defined as a density larger than 1cm) represents a confluence of the individual silica granulomas. These masses usually begin in the peripheral upper lobe and typically have a sharp lateral margin with a clear zone of over-inflated emphysematous spaces between the lesion and the chest wall. The masses are often bilateral, symmetric, and have a diameter of over 1-2 cm [5,7]. The masses retract toward the hilum over time and although they may occasionally calcify, they rarely cavitate. Egg-shell calcification of hilar nodes in found in only 5% of cases (and may also be seen in Sarcoid, TB, Histoplasmosis, and treated lymphoma).
Computed tomography: A number of parenchymal features may
be identified on CT/HRCT and include:
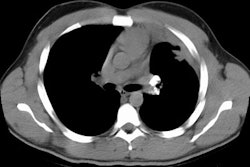
1- Nodules: The most characteristic finding is multiple small,
well-defined nodules 2-5 mm in diameter most numerous in the upper
lobes [7]. The nodules can appear as round or branching
structures. The nodules are observed in centrilobular, paraseptal,
and subpleural regions and have a perilymphatic distribution [7].
These micronodules
correspond to irregular fibrosis surrounding respiratory
bronchioles
and they may be difficult to distinguish from normal vascular
markings.
2- Emphysematous change: Contraction of a nodule results
in a small, surrounding halo of emphysema to form.
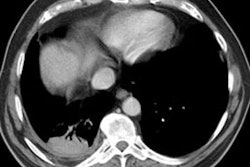
3- Conglomerate
masses: These lesions appear as irregularly marginated masses,
larger than 2 cm in size. They converge towards the hila and leave
behind peripheral emphysema. These masses can be indistinguishable
from lung carcinoma or TB. Parenchymal bands can be noted in
association
with congomerate masses. Conglomerate masses are often
hypermetabolic on FDG PET imaging [9].
Nodular septal thickening may also be seen in patients with silicosis, but intralobular interstitial thickening is uncommon (a pattern typical of UIP occurs in only about 10% of silicosis patients [9]).
Hilar and mediastinal lymph node enlargement may precede the appearance of parenchymal nodular lesions [7]. Nodal calcification is common and typically occurs at the periphery of the node (egg-shell calcification) [7].
In more advanced disease pleural abnormalities such as pleural effusion and pleural thickening (general adjacent to the area of progressive massive fibrosis) can also be seen [6].
REFERENCES:
(1) AJR 1992; Acute silicosis. Case of the day. 158: 1361-62
(5) Radiographics 2001; Kim KI, et al. Imaging of occupational lung disease. 21: 1371-1391
(6) Radiololgy 2005; Arakawa H, et al. Pleural disease in silicosis: pleural thickening, effusion, and invagination. 236: 685-693
(7) Radiographics 2006; Chong S, et al. Pneumoconiosis: comparison of imaging and pathologic findings. 26: 59-77
(8) AJR 2007; Marchiori E, et al. Silicoproteinosis:
high-resolution CT findings in 13 patients. 189: 1402-1406
(9) Radiology 2014; State of the art: imaging of occupational
lung disease. 270: 681-696