Siemens Healthineers has received U.S. Food and Drug Administration (FDA) 510(k) clearance for six new interventional radiology systems in its Artis portfolio, all featuring the Optiq AI imaging chain.
The cleared systems include floor, biplane, ceiling, and robotic floor-mounted versions of the Artis vision platform, as well as floor configurations of the Artis icono.explore and Artis genio platforms, according to Siemens. The vendor first showcased the new platforms at RSNA 2025.
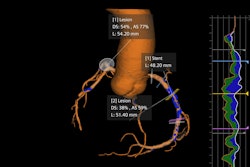
All include Optiq AI, which applies deep–learning-based noise reduction across 2D imaging modes such as fluoroscopy, acquisition, and digital subtraction angiography, the company said.
Siemens noted additional capabilities across the portfolio, including the following:
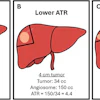
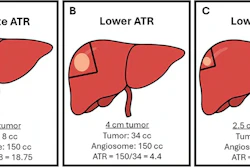
- syngo DynaCT MORE, a 3D application that reduces motion-related artifacts to enhance intraprocedural 3D imaging.
- Touch UI, a touchscreen enabling tableside access to key system functions and physician/technologist collaboration.
- Standardized controls across all six platforms to reduce variability when teams move between rooms.
 Artis for interventional imaging
Artis for interventional imaging