Bronchogenic Carcinoma:
Click here for a Discussion of post-pneumonectomy complications
Click here for a Discussion of staging bronchogenic carcinoma
Click here for a Discussion of Helical CT screening for lung cancer
Click here for a Discussion of PET imaging in lung cancer
View cases of lung cancer (Many more images in section on Staging lung cancer)
Clinical:
Bronchogenic carcinoma is the most common malignancy and most
common cause of cancer related deaths in both men and women. About
85% of lung cancer cases are associated with cigarette smoking,
however, only 15% of smokers ultimately develop lung cancer [39].
Approximately 20% of women who develop lung cancer are non-smokers
[78].
Sputum cytology is occasionally used to make the diagnosis of bronchogenic carcinoma. It is most likely to be positive in patients with squamous cell carcinoma, large tumors, or those that are central in location. Specimens are best obtained as early morning or with induced sputum samples. The incidence of synchronous multiple primary tumors is under 3.5% [34]. Risk factors for lung cancer include:
1- Cigarette smoking: Confers a 4 to 120 times increased risk for bronchogenic carcinoma (15-30 x's in heavy smokers). The risk for developing cancer is related to the number of pack-years of smoking [39]. Some suggest that the risk returns to normal 13 years following cessation of smoking, but others say that the risk always remains higher than that of non-smokers. Also- there is a statistically significant association between obstructive ventilatory impairment and lung cancer [63]. Smokers with reduced FEV1/FVC ratios have a higher risk of acquiring lung cancer (particularly airways lesions) compared to smokers without a ventilatory impairment [63].
2- Asbestos: Confers a 4 to 5 times increased risk, but up to a 100 times increase risk if exposed individual is also a smoker.
3- History of Head and Neck Cancer
4- History of Interstitial Lung Disease (Up to 6-12% of patients with idiopathic pulmonary fibrosis will develop bronchogenic carcinoma, most commonly adenocarcinoma)
5- Exposure to toxic agents: Arsenic, Nickel (squamous cell), Chromium, Chloromethyl ether (small cell carcinoma)
6- Uranium exposure (small cell carcinoma)
7- Prior lung cancer: The longer an individual survives following primary resection of a lung cancer, the greater their chance of manifesting a subsequent primary lung cancer [53]. The risk for subsequent lung cancer has been estimated to be cumulative and on the order of 1-3% per year [53]. Between 10-32% of patients surviving resection for lung cancer may develop a second primary lung tumor over a 10 year period of time [34,53].
8- HIV: Patients with HIV are likely at an
increased risk (6.5 fold increased incidence [14]) for developing
bronchogenic carcinoma- especially HIV positive patients with a
history of smoking. Patients present at a significantly lower age
(49 years) than the general population (68 years) [14]. There is
no significant difference between lung cancer cell types in HIV
patients and their non-HIV counterparts (with adenocarcinoma being
the most common followed by squamous cell and small cell [96] ,
however, most patients present with stage II or IV disease [96]
and there does appear to be a high incidence of rapid tumor
progression with local and distant metastases in HIV individuals.
The diagnosis of lung cancer may precede the diagnosis of HIV
infection. [1,2,14].
The radiographic appearance is similar to that of bronchogenic carcinoma in the general population. The increased incidence of neoplasms in HIV-patients may be related to increased life expectancy in these individuals now that there is better management of opportunistic infections.
9- Radon exposure [26,78] - exposure to ionizing radiation from radon is the second leading cause of lung cancer mortality in the US [78]. The EPA calculates that radon is responsible for 21,000 lung cancer deaths per year and that 2,900 of these deaths occur in patients who were never smokers [78].
10- Second hand smoke- second hand smoke exposure is associated with an increased incidence of lung cancer [78].
11- Family history- patients with a family history of lung cancer have an increased incidence of lung cancer- even in non-smoking families [78].
12- Human papilloma virus infection- HPV infection has been associated with an increased incidence of lung cancer in non-smoking women in Asia [78].
Previously, squamous cell carcinoma accounted for 30-40% of cases of bronchogenic carcinoma and was roughly as common as adenocarcinoma. However, recent data suggests that squamous cell cancer now accounts for only 23% of NSCLC cases [77]. The lesion is generally central (70% hilar or perihilar in subsegmental or larger bronchi) and there is a strong association with cigarette smoking. It is the most likely bronchogenic carcinoma to cavitate- about 15% bronchogenic carcinomas are cavitary, and of these, nearly 60% are squamous cell lesions [ACR Syllabi #40:p.92]. The wall of the cavity is typically thick and nodular. Squamous cell carcinomas often demonstrate an intralumenal growth pattern- often resulting in distal atelectasis or post-obstructive pneumonitis (a non-infectious process). Peripheral squamous cell tumors frequently show a desmoplastic response to tumor growth within the adjacent lung [3]. SCCa has the lowest frequency of distant metastases which are present in under 20% of cases at the time of diagnosis. More often the tumor spreads to involve local nodes by direct extension. Squamous cell carcinoma of the lung has the most favorable prognosis of all bronchogenic carcinomas. Due to an ability to produce a parathormone-like substance, SCCa is the most common bronchogenic carcinoma to cause hypercalcemia. Hypertrophic osteoarthropathy is also seen most commonly in association with this cell type.
Pancoast's tumor: A Pancoast's tumor (superior sulcus tumor) is a rare form (1%) of bronchogenic carcinoma which arises in the lung apex. It is most commonly of the squamous cell type, but adenocarcinomas or large cell carcinomas can also occur in this location. The Pancoast syndrome results from invasion of the pleura and rib, producing shoulder pain; involvement of the brachial plexus causing arm pain and paresthesias (particularly in the ulnar nerve distribution); and spread to the sympathetic stellate ganglion leading to Horner's syndrome. Symptoms of Horner's syndrome include ptosis of the upper eyelid, myosis, and anhydrosis; as well as constriction of the pupil, enophthalmos (sinking in of the eyeball), narrowing of the palpebral fissure, and flushing of the affected side of the face. Patients with Horner's syndrome have a worse prognosis, but this is not a contraindication to surgery. Patients with limited chest wall invasion and no evidence of distant metastases are considered potentially curable (Stage IIB). MRI is more accurate than CT in determining the extent of chest wall invasion. The treatment of choice is radiation therapy followed by surgery- or radiation therapy alone for patients with unresectable lesions. The following criteria usually indicate an unresectable lesion: tumor invasion of the great vessels at the thoracic inlet- most commonly the subclavian artery because of it's location; phrenic or recurrent laryngeal nerve paralysis; invasion of the vertebral bodies, trachea, or esophagus. Resection of the lower cords of the brachial plexus (if involved) may be necessary, but extensive involvement usually renders the patient inoperable. Preoperative XRT is given to reduce tumor size. Typically 30-45 Gy is administered 3 to 6 weeks prior to surgery. Surgery involves en bloc resection of the chest wall, posterior portions of the first three ribs, portions of the upper thoracic vertebral bodies (including the transverse processes), the intercostal nerves, the lower trunk of the brachial plexus, the stellate ganglion, a portion of the dorsal sympathetic chain, and a lobectomy of the involved lung. Surgery is associated with a high morbidity (7-34%) and mortality (3-10%). [ACR Syllabus #40:p.295-312]
Adenocarcinoma is now more common than squamous cell carcinoma
(43% of cases of NSCLC) [77] and the incidence of adenocarcinoma
has been increasing over the past few decades [72]. The increased
incidence has been attributed to the use of filtered, low tar,
nicotine cigarettes that results in deposition of carcinogens in
the peripheral lung parenchyma [77]. Lung adenocarcinoma
represents a variety of histologic subtypes and usually tumors
contain two or more histologic patterns [85].
The lesion is generally peripheral (75%) and may be associated with a scar (the scar may actually represent desmoplastic reaction to the tumor). It is the most common cell type to be found in non-smokers, however, most affected patients are still smokers. It is thought that smokers of low tar cigarettes inhale more deeply and retain smoke in more peripheral portions of the lung and thus acquire peripheral adenocarcinomas [63]. Adenocarcinomas uncommonly cavitate and should calcification be present within the tumor, it is typically related to the presence of a pre-existing granuloma which has been engulfed by the tumor. The tumor commonly metastasizes early to lymph nodes, the pleura, adrenal glands, CNS, and bone. It has been shown that adenocarcinomas, including those with BAC features, disproportionately respond to treatment with tyrosine kinase inhibitors [72].
Atypical adenomatous hyperplasia (AAH) is a pre-invasive lung
lesion that typically appears of ground glass attenuation and
measures less than 5 mm on CT imaging [86].
Because the previous term bronchoalveolar carcinoma included
several subcategories of varying histopathologic, radiologic, and
prognostic clinical importance, the histologic classification of
adenocarcinoma of the lung has been revised [86]. The new
classification system includes four new terms:
1. Adenocarcinoma in situ (AIS- formally BAC): A small (less than
or equal to 3 cm) tumor with purely noninvasive lepidic growth
(along the alveolar walls without stromal, vascular, or pleural
invasion) that typically appears non-solid (ground glass) on CT
[86,87,117]. The lesion arises from Clara cells and/or type II
pneumocytes [87]. If the lesion is resected, patients have a 100%
disease-free survival [86].
2. Minimally invasive adenocarcinoma (MIA): A small (less than or
equal to 3 cm) tumor with predominantly lepidic growth that has a
focus of invasion that measures 5 mm or less in size [86].
Following resection, these patients likely have a 100% disease
free survival [86,117]. The lesion typically appears of ground
glass attenuation on CT, but may have a small solid component that
measures up to 5 mm [86].
3. Lepidic predominantly non-mucinous adenocarcinoma: An invasive
nonmucinous adenocarcinoma that shows a predominant lepidic growth
pattern, but has an invasive component larger than 5 mm [86]. On
CT, the lesion typically appears part solid (solid component
larger than 5 mm), but it may also appear nonsolid or occasionally
have cystic components [86,117].
4- Invasive adenocarcinoma defined based on the predominant
histologic pattern:
- Non-mucinous (previously mixed type
adenocarcinoma): Subclassificaitons have been added to the
non-mucinous category to reflect various histologies and includes
lepidic predominant (formally non-mucinous BAC pattern), acinar
(intermediate prognosis [88]), papillary (intermediate prognosis
[88]), micropapillary, or solid predominant [86,87].
- Micropapillary is a new subtype
and is associated with a less favorable prognosis (5-year survival
for lesions equal or less than 20mm is 54%, compared to 81% for
nonmicropapillary tumors) [86].
- Solid predominant is also associated with a poor prognosis in
early stage adenocarcinoma [86,88].
- Invasive mucinous adenocarcinoma
(formally mucinous BAC): Invasive mucinous adenocarcinoma with
lepidic growth as its predominent component [86]. This variant has
a distinctive histologic appearance with tumor cells having a
goblet or columnar cell morphology with abundant intracytoplasmic
mucin [87]. It can have a variable radiologic appearance ranging
from mostly solid to part-solid or non-solid, or appear as a
pneumonic infiltrate [86,87]. The lesion can be single or commonly
multifocal (reflecting aerogeneous spread) [86,87]. KRAS mutations
can be found in up to 80-100% of invasive mucinous
adenocarcinomas, but are found much less frequently in
non-mucinous tumors [86]. The lesion is associated with an overall
worse prognosis [88].
- Other histologies include colloid
adenocarcinoma (worse prognosis [88]), fetal adenocarcinoma, and
enteric differentiation [87].
The new histologic classification also notes that growth rates
for adenocarcinoma of the lung are not necessarily constant and
that cases have been described in which followup CT may reveal a
slight decrease in tumor volume (presumably due to retraction
caused by alveolar collapse and fibrosis) [86]. Also, for small,
solitary, early-stage tumors, the size of the invasive component
is an independent predictor of survival [86]. In patients with
pure GGNs, a lesion size less than 10 mm is very suggestive of a
pre-invasive lesion [91]. The five year survival rates for AIS and
MIA are nearly 100%, but decrease to 90%, 83%, and 84% for
lepidic, papillary, and acinar predominant stage I non mucinous
adenocarcinoma, respectively [113]. Patients with node negative
invasive adenocarcinomas appear to be at an increased risk for CNS
metastases compared to other lung cancers [86]. For node negative
patients, a 2 cm primary lesion has a 14% probability for CNS
metastases, and a 6 cm lesion has a 64% probability [86].
The presence of EGFR mutations in nonmucinous adenocarcinomas is
predictive of a therapeutic responsiveness to EGFR tyrosine kinase
inhibitors (such as gefitinib) [86,87]. These mutations are more
common in women than men, in non-smokers compared to smokers, in
persons of East Asian ethnicity, and in well to moderately
differentiated tumors (compared to poorly differentiated lesions)
[86].
Bronchoalveolar cell carcinoma (BAC):
This is an older discussion of BAC. See the above discussion for adenocarcinoma for the new classification of what was formerly BAC.
Bronchoalveolar cell carcinoma is a highly differentiated subtype of adenocarcinoma that accounts for 1.5-6.5% of all pulmonary neoplasms [12,72]. Although BAC has the weakest association with smoking, between 25-50% (up to 81% [17]) of affected patients still have a smoking history. There is a strong association with underlying interstitial lung disease, parenchymal scarring (found in up to 27% of cases), and exogenous lipoid pneumonia. The lesion also has a higher incidence in women and has a younger age distribution [72. BAC demonstrates a unique growth pattern spreading along the alveolar septa without stromal invasion and typically has an indolent course [72]. The lesion may appear stable for a period of 2 to 7 years before the diagnosis of BAC is made [17]. Compared to other forms of lung cancer, BAC also exhibits a relatively high incidence of multifocality (25% versus 5%) [72]. Limited surgical resection is an option for patients with minimally invasive adenocarcioma and part-solid nodules with greater than 50% ground glass component [72]. In these cases, radical segmentectomies with hilar and mediastinal lymph node dissection or sampling only still results in high curability rates [72].
Histologically, two subtypes of BAC are identified- mucinous and non-mucinous. The mucinous form probably arises from columar mucous-containing cells, is more common (about 80% of cases), is more likely to be multicentric, may result in a false negative result on PET FDG imaging, and has a worse prognosis. Patients affected with the mucinous subtype can present with massive bronchorrhea, although this occurs in only about 5% of cases [12]. The non-mucinous form probably arises from type 2 pneumocytes or Clara cells, is more likely to be localized, and has a better prognosis. BAC characteristically grows along the pulmonary interstitium without destroying lung parenchyma (referred to as "lepidic growth"). It has been suggested that BAC with pure lepidic growth be classified as adenocarcinoma in situ due to the very favorable prognosis [85]. The tumor may spread by way of the tracheobronchial tree to other sites within the ipsilateral or contralateral lung (aerogenous spread). When this occurs, there may be a focal area of consolidation within one lung associated with bilateral pulmonary nodules. [4] The presence of aerogenous spread is associated with a worse prognosis [17]. The presence of vascular invasion identified at histologic analysis is also associated with a worse prognosis [17].
Bronchoalveolar cell carcinoma may present in a number of ways- a solitary peripheral nodule (SPN) is the most common presentation and occurs in about 45% of cases (but up to 85% of cases can present as an SPN[17]). Lesions that present as a solitary nodule are associated with the best prognosis, especially if the lesion is less than 3 cm in size. Other presentations which are associated with a worse prognosis include a localized or diffuse indolent consolidation (about 30% of cases) [22], or multiple nodules (about 25% of cases)- most commonly too numerous to count acinar sized nodules. The nodules can range in size from 1 mm to 3 cm, can be sharp or poorly defined, can show a halo sign, can cavitate, and can have associated regions of confluence/consolidation or ground-glass opacity [27].
BAC commonly presents as a focal area of ground-glass opacification [5,61]. Ground-glass attenuation about the margins of a semisolid lesion in other cases is felt to be due to the lepidic growth of the tumor [12]. The greater the ground-glass component of a lung cancer, the more likely the lesion will behave in an indolent manner and the better the prognosis (particularly for peripheral adenocarcinomas smaller than 3cm) [61,84]. In one study, for lung adenocarcinomas smaller than 2 cm with more than 50% GGO, there was 0% incidenc of lymph node metasatases [84]. Additionally, for adenocarcinomas, the greater the degree of BAC component, the better the overall prognosis [72]. Cystic change (pseudocavitation) may uncommonly be seen in association with BAC. Rather than representing true cavitation of the tumor, this finding most likely represents cystic or traction bronchiectatic changes within adjacent scarred or fibrotic lung. Pleural effusion is found in about a third of cases, as is the presence of hilar or mediastinal adenopathy. The "CT angiogram sign" can be seen and refers to an area of low density consolidation (due to the fluid and mucous produced by the tumor) in which prominent enhancement of the pulmonary vessels is seen following the administration of intravenous contrast. This finding is non-specific, however, as it can also be found in cases of post-obstructive consolidations, lobar pneumonia, pulmonary lymphoma, extrinsic lipoid pneumonia, pulmonary infarction, passive atelectasis, and pulmonary edema [12,16,23].
Mucinous bronchoalveolar carcinoma is a subtype of BAC which may demonstrate diffuse, heterogeneous or homogeneous low attenuation (below 10 HU) and only moderate contrast enhancement. A "crazy-paving" appearance in which a patchy or geographic ground-glass opacification is superimposed upon smooth septal thickening has also been described [6]. This pattern is also seen in patients with alveolar proteinosis, but in these cases it is usually bilateral and symmetric. [7]
|
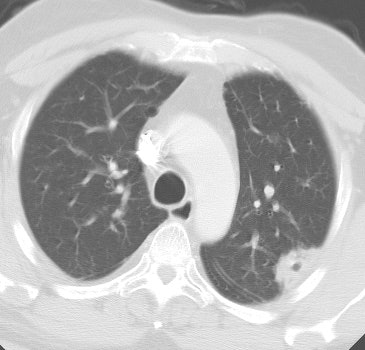
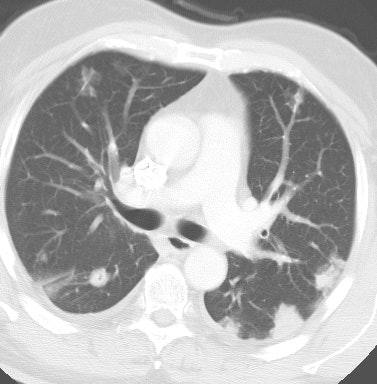
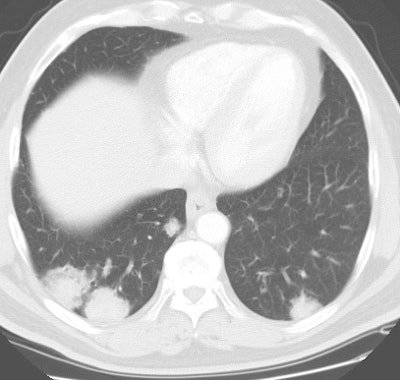
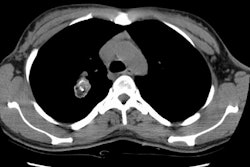
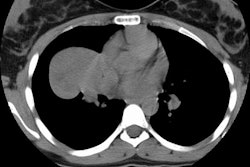
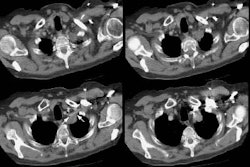
Bronchoalveolar cell carcinoma presenting at multiple poorly marginated lung nodules- some of which appear cavitary. The findings mimic a pulmonary infectious process. |
|
|
Small cell carcinoma accounts for 15-20% of primary lung
malignancies and has the strongest association with cigarette
smoking (cigarette smoking is responsible for 95% of cases [97]).
Small cell lung cancer is a neuroendocrine neoplasm with the
highest mitotic rate (more than 10 mitoses per HPF) and it is
considered to be the most aggressive form of lung cancer. The most
common subtype of small cell carcinoma is the oat cell variety.
About 20% of small cell carcinomas will contain elements of
non-small cell histology, most commonly squamous cell carcinoma.
Small cell carcinoma is the most likely to produce ectopic
hormones- most commonly resulting in syndrome of inappropriate
antidiuretic hormone (SIADH- characterized by decreased urine
concentration, reduced plasma osmolarity, and euvolemic
hyponatremia), but Cushings syndrome (ACTH) or acromegaly (due to
ectopic growth hormone releasing peptide) can also occur [97].
Neurologic paraneoplastic syndromes include autoimmune neuropathy
(Eaton-Lambert syndrome), encephalomyelitis, and limbic
encephalitis (and neurologic symptoms can precede the diagnosis of
lung cancer by up to 2 years and can improve after treatment of
the cancer) [97].
The lesion is generally central (85-90% within a lobar or
mainstem bronchi) and has a tendency to invade longitudinally
along the bronchial wall, in a submucosal and intramural fashion
[3]. About 5% to 10% of patients have a solitary pulmonary nodule
[55]. Internal necrosis is common, but cavitation is extremely
rare. Often times, the primary lesion is not evident and the
predominant finding is metastatic hilar (seen in 84% of cases) and
mediastinal adenopathy (92% of cases) [55,70]. As a result of this
adenopathy, small cell carcinoma is the most common bronchogenic
carcinoma to cause superior vena caval syndrome. Intratumoral
calcification has been reported in up to 23% of patients [97].
Small cell carcinoma has previously been classified using a two stage system as either extensive or limited disease [28]. Limited disease has been defined as disease limited to a single hemithorax (although local extension of the primary tumor and mediastinal/supraclavicular adenopathy could be present if it could be included in the same radiation port as the primary tumor) [74,92]. It has also been defined as tumor confined to one hemithorax plus regional hilar or mediastinal lymph nodes (including contralateral mediastinal nodes), or supraclavicular nodal metastases, but no cervical or axillary adenopathy, and ipsilateral pleural effusion (either cytologically positive or negative) [28,75]. Extensive disease defines all other patients including the presence of a malignant pleural effusion and disease beyond a single radiation port [92]. However, "limited disease" made no distinction between a small intraparenchymal SCLC and a large mass invading the mediastinum with multiple ipsilateral nodal metastases [74]. As such, it has been recommended that the standard lung cancer TNM staging system be applied to small cell lung cancer [74,75]. I this system, stages I-III correspond to limited stage and stage IV to extensive disease [97]. Using the TNM staging system, the reported 5 year survival for patients with small cell lung cancer are 56% for stage IA, 57% for stage IB, 38% for stage IIA, 40% for stage IIB, 12% for stage IIIA, and 0% for stage IIIB [75].
About 60-70% of patients have extensive (disseminated/metastatic)
disease at presentation [66,97]. Common sites of metastatic
disease at time of diagnosis include the bones (19-38% of
patients), liver (17-34%), adrenal glands (10-17%), and the brain
(up to 14% and MR imaging of the brain is recommended in all
patients) [66,97]. A malignant pleural effusion can be seen in
10-38% of patients [101]. The presence of even a minimal pleural
effusion (< 10 mm thick) can be seen in up to 21% of patients
and its presence is associated with a worse prognosis [101].
FDG PET imaging can play an important role in the evaluation of patients with small cell lung cancer [51,55]. Compared to conventional imaging, FDG PET can result in upstaging from limited to extensive disease in up to 19% of patients [97]. Findings on PET imaging can lead to a change in initial management in up to 27% of patients [97].
Small cell carcinoma is associated with the worst prognosis,
despite typically good response to initial chemotherapy the
majority of patients suffer a relapse within 2 years [4,97]. A
combination of concurrent chemotherapy and radiation is used for
treatment of limited stage disease, but is curative (5 year
survival) in only about 10-20% of patients [28,35,97]. Extensive
stage disease is treated with chemotherapy alone and has a median
survival of nine months and a 5 year survival of 1-2% [35,97].
Prophylactic cranial irradiation can reduce the incidence of brain
metastases by nearly 50% and has been associated with increased
3-year survival rates [97].
Large cell neuroendocrine carcinoma: The tumor is a poorly differentiated and high grade neuroendocrine tumor that is morphologically and biologically between an atypical carcinoid tumor and small cell lung cancer [55]. The lesion accounts for about 3% of resected primary lung cancers [55]. The lesion usually appears as a well-defined, lobulated mass with no air bronchograms or calcifications [55].
4. Large Cell Neuroendocrine Carcinoma:
Large cell carcinoma accounts for only 5-10% of primary lung malignancies and is strongly associated with cigarette smoking. The lesion is typically peripheral and generally large (over 4 to 6 cm), with rapid growth, early metastases, and a poor prognosis. Giant cell carcinoma is a subtype of large cell carcinoma which is highly malignant and associated with a very poor prognosis.
Paraneoplastic syndromes associated with bronchogenic carcinoma:
1. Hypercalcemia: Most commonly associated with squamous cell carcinoma due to secretion of a parathormone like substance. Hypercalcemia has also been described in association with renal cell, hepatoma, and pancreatic carcinoma.
2. Ectopic ACTH Production: Most commonly associated with small cell carcinoma. Only 10% of affected patient's will present with manifestations of Cushing's syndrome.
3. SIADH: Syndrome of inappropriate secretion of anti-diuretic hormone. The SIADH syndrome occurs in only 1-2% of patients with bronchogenic carcinoma. It is most commonly associated with small cell carcinoma. The result of continued ADH secretion is increased resorption of water in the renal tubules which results in water retention and hyponatremia. Symptoms of confusion, irritability, and weakness occur when the serum sodium level decreases below 120 meq/L.
4. Eaton-Lambert syndrome: A peripheral neuropathy with myasthenia-like symptoms.
5. Acanthosis nigrans
6. Hypertrophic Osteoarthropathy (HOA): Hypertrophic osteoarthropathy occurs in up to 10% of patients with bronchogenic carcinoma and consists of 2 components: 1- Clubbing of the fingers & toes; and 2- Periostitis of the long bones. Patients may have pain and swelling about the wrists, & less commonly the ankles and knees. Plain film findings in HPOA reveal a thick, lamellar periosteal new bone formation. Bone scan may show patchy or linear increased activity along the periosteum of long bones. HPOA tends to resolve following tumor resection. HPOA can also be seen in: Bronchiectasis, cystic fibrosis, mesothelioma, pneumoconiosis, cyanotic heart disease, and inflammatory bowel disease.
7. Anti-Hu paraneoplastic syndrome (Encephalomyelitis or sensory neuropathy): Patient's with this syndrome may have either an encephalomyelitis with symptoms such as confusion, memory loss, dementia, ataxia, tremor, diplopia, dysarthria, and dysphagia; or a subacute sensory neuropathy with asymmetric paresthesias. A specific antineuronal antibody (anti-Hu) is found in the CSF and serum of affected patients. Nearly 90% of affected patients have localized small cell lung cancer, but the syndrome occurs in less than 1% of patients with small cell lung cancer. Interestingly, the syndrome often begins prior to the diagnosis of cancer. Lack of findings on plain radiographs does not exclude a lung cancer in patients with anti-Hu syndrome and chest CT should be performed to look for occult neoplasm. [20]
Genetics and newer chemotherapeutic agents:
EFGR-mutant non small cell lung cancer:
Epidermal growth factor receptor (EFGR) is a transmembranous receptor tyrokinase involved in signaling pathways that regulate cell proliferation, apoptosis, angiogenesis, and invasion [94,112]. Activating mutations of EGFR are present in between 15-50% of patients with lung adenocarcinomas [94,99]. EGFR mutations are more common in Asians. female patients, and never smokers with adenocarcinoma histology, and are less common in older patients with a significant smoking history [94,99,107,112]. People of Asian descent also have higher response rates to EGFR inhibitors [99]. Several types of EGFR mutations are considered sensitizing mutations with a response to EGFR-TKI treatment including exon 19 deletions or L858R point mutation in exon 21 [94].
EGFR kinase inhibitor agents (EGFR-TKI) that are currently being used are bevacizumab, erlotinib (Tarceva), afatinib (Gilotrif), and in Europe gefitinib (Iressa) [71,76,94,99]. Typically, adenocaricnoma patients with East Asian ethnicity, female sex, and non-smoker status respond well fo EFGR-TKI therapy [71,110]. The presence of K-ras mutations is associated with resistance to these drugs [71].
EGFR mutation positive patients (EFGR-mut) have higher response
rates than EGFR wild-type (EGFR-wt) patients [109]. EGFR tyrosine
kinase inhibitors have initial response rates in excess of 70% and
are associated with progression free survival of 9.7-13.1 months
in selected patients [94,99,103]. Unfortunately, 10-30% of
patients present with primary resistance, and 50-100% of patients
develop acquired resistance within 9-12 months [99,107,110].
Additionally, approximately 10-20% of patients who have a partial
response to EGFR-TKIs do not posses EGFR mutations [110]. Other
factors associated with poor response and shortened progression
free survival include the presence of a pleural effusion, N2 or N3
disease, and the presence of distant metastases [101]. In patients
that develop resistance to erlotinib, the agent is typically
continued because of a flare phenomena with accelerated disease
progression if the agent is stopped [99]. Studies have
demonstrated that TKI-sensitive tumor cells down-regulate glucose
uptake early after exposure to TKI treatment and tumor volume
metrics (TLG) may be better able to identify non-responding
patients compared to SULmax [109].
Bevacizumab is recommended with chemotherapy for the NSCLC
(adenocarcinoma and large cell carcinoma) without a recent history
of hemoptysis [98]. The agent is not used with squamous cell lung
cancer due to potential for life-threatening hemorrhage [98].
Bevacizumab causes central tumor necorsis with tumor cavitation
indicating response to treatment (cavitation occurs in 14-24% of
patients) [98]. Erlotinib inhibits the function of the epidermal
growth factor receptor, and bevacizumab targets the circulating
vascular endothelial growth factor [76]. The resultant effects is
supposed to decrease vascular permeability, microvascular density,
and cell density that eventually result in tumor necrosis [76].
Gefitinib has a reported response rate of approximately 74% and
can extend overall survival times for patients with EGFR mutation
by approximately 7 months compared to standard chemotherapy [107].
The therapy often results in consolidation rather than mass regression leading to limitations with conventional CT imaging for the determination of tumor response [76]. Tumor cavitation, rather than decreased tumor size, is another common finding in NSCLC treated with vascular EGFR inhibitors (up to 24% of patients) [80]. Because the cavitary portion of the tumor does not contribute to tumor volume, the assessment of post treatment tumor burden may be improved by incorporation of the cavitation into the tumor measurement [80].
Pneumonitis and interstitial lung disease are among the more
common toxicities associated with EFGR-TKI agents such as
gefitinib and erlotinib [112]. Several different patterns of
pulmonary toxicity have been reported including NSIP, COP, and AIP
[112].
Anaplastic lymphoma kinase (ALK) mutant non-small cell lung
cancer:
The anaplastic lymphoma kinase (ALK) rearranged oncogene is
another molecular target present in 2-7% of NSCLC patients (more
commonly younger patients, women, light or never smokers, and
adenocarcinoma histology) [94,112]. Crizotinib (Xalkori) is an
adenosine triphosphate competitive inhibitor of the ALK receptor
tyrosine kinase that has a reported overall response rate of 57%,
a PFS of 8 months, and ore than 50% survival at two years after
starting treatment [94].
First line treatment includes crizotinib, which is an ATP
competitive inhibitor of the ALK receptor tyrosine kinase [112].
The agent has an overall response rate of more than 50% and leads
to PFS of 8 months in ALK positive patients [112]. If resistance
develops, 2nd and 3rd generation agents can be used including
ceritinib, alectinib (also has CNS penetrance which can aid in
reducing risk for brain mets), brigatinib, or lorlatinib [112].
Side effects associated with crizotinib include pneumonitis and
ILD, as well as hepatotoxicity, neutropenia, and CNS relapses
[112]. Approximately 20% of patients undergoing crizotinib
treatment will develop either new or more complex appearing renal
cystic lesions [112]. These cystic lesions tend to regress after
cessation of crizotinib [112]. Osteopenia, often leading to
compression fratures, is another common complication [112].
Alectinib has also been associated with pneumonitis and ILD
[112].
KRAS mutated NSCLC:
Approximately 25% of pulmonary adenocarcinomas are KRAS positive
[106]. Tumors with the KRAS mutation have been found to respond
poorly to both tyrosine kinase inhibitors and conventional
cisplatin-based chemotherapy [105].
Approximately 5% pf patients with NSCLC have a chomosomal
rearrangement that causes hyperactivation of ALK [99]. ALK
rearrangements occur most commonly in patients that never smoked
or light smokers [99]. Patients also tend to be younger than 65
years of age [99]. There is no ethnic association [99]. Crizotinib
is an oral ALK inhibitor with a 60% response rate in patients with
EML4-ALK rearrangement [99].
BRAF mutation is found in 3-5% of adenocarcinomas and it is
predominantly seen in current or former smokers [107]. Mutation in
BRAF V600E has been shown to result in aggressive histologic
findings and decreased disease-free and overall survival rates
compared to non-V600E mutations [107].
FDG PET imaging can be used to demonstrate decreased metabolic
function consistent with tumor treatment even if the lesion is not
changing in size [76]. In one study, a decrease of more than 20%
in tumor glucose metabolism at 3 weeks following initiation of
treatment was associated with a favorable progression free
survival time- regardless of any change in tumor size [76].
However, another study demonstrated that a tumor response can be
seen as early as one week following initiation of therapy with
erlotinib [95]. Another study suggested that greater percentage
decrease in SUV max was associated with a higher response rate and
decreased tumor heterogeneity on FDG PET imaging 6 weeks following
erlotinib therapy was associated with longer patient survival
[100].
NOTE: Potential life-threatening hemorrhage may occur in patients
with squamous cell carcinoma who receive bevacizumab.
Monoclonal antibody therapy targeting immune checkpoints (ICI
therapy):
Programmed-death 1 (PD-1) and Programmed-death ligand 1 (PD L1)
therapy restore antitumor T-cell activity and can prolong survival
in certain NSCLCs [115]. Patients with NSCLC whose tumor
demonstrates at least 50% PD-L1 expression have a response rate of
45% to PD-1 directed treatment [114]. However, even patients
without PD-L1 expression still have a 10% chance of obtaining a
response [114]. The limited PPV of PD-L1 immunohistochemistry
might be caused by treatment resistance mechanisms involving
pathways beyond the PD-1-PD-L1 axis, such as insufficient tumor
antigenicity, impaired antigen presentation, or an
immunosuppressive microenvironment [114].
Pseudoprogression has been described, in which the anticancer
immune reaction activated by immune checkpoint inhibitor treatment
may initially increase the total tumor volume because of
inflammatory cell infiltration or result in the appearance of new
lesions [116]. The rate of pseudoprogession in NSCLC patients
treated with ICIs has been reported to be 0% to 6% [116].
However, in a subset of patients, there is growing evidence that
treatment with immune checkpoint inhibitors can have a detrimental
effect characterized by a rapid increase in tumor extent with a
worse clinical outcome- this is referred to as hyperprogressive
disease (HPD) [115]. The prevalence of HPD varies from 4% to 29%
[115]. NSCLC patients with high metabolic tumor burdens on PET
imaging (expressed by MTV and TLG), may be at increased risk for
HPD following initiation of immune checkpoint inhibitor therapy
[115]. Other factors that may be associated with an increased risk
for HPD include age over 66 years and female sex [115].
Alternative therapy:
1- Percutaneous radiofrequency ablation:
Percutaneous radiofrequency ablation may offer an alternative for
patients that are not surgical candidates or those with
unresectable tumors and is generally well tolerated
[54,57,59,62,67]. The benefits of this technique include
preservation of more lung tissue and a reduction in morbidity
compared to surgical resection [60]. RF ablation does not induce
significant changes in lung function and spirometry results [67].
In RFA, a high frequency alternating current is applied via an
electrode needle that has been placed within the tumor [81]. This
heats the surrounding tissue 60-100?C creating a necrotic zone and
destroying tumor cells [81,108]. Lung tumors are well suited to
RFA because the surrounding air in the adjacent lung parenchyma
provides an insulating effect helping to concentrate the
radiofrequency energy [81]. Factors that limit RF in the lung are
the heat-sink effect caused by adjacent blood vessles larger than
3 mm in diameter that produce a cooling effect through flowing
blood around the tumor and, as mentioned previously, the air
present in the ventilated lung is refractory to electrical and
thermal conduction [90,108].
Tumor necrosis of greater than 90% can commonly be achieved [62].
Smaller lesions (less than 3 cm) are more likely to have
demonstrate complete necrosis following therapy [57,59] which is
associated with a better long term survival [57,79]. The intended
ablation should encompass the entire tumor and a 1 cm rim of
normal tissue as a safety margin to ensure complete ablation
[58,81]. Other authors report that an ablation diameter twice that
of the tumor diameter improves the success rate [67], while other
indicate that the target ablation diameter should be 1.6-2 cm
greater than the tumor diameter (in order to decrease the risk of
local recurrence) [73]. A ratio of RFA-induced GGO to tumor area
of greater than 4 is another criteria that has been associated
with a significantly higher rate of complete ablation [108]. An
ablation margin of less than 4.5mm or an incomplete margin has
been correlated with local tumor progression [108].
RFA has been shown to offer a survival benefit for patients with
lung tumors with a 3 year survival range of 46-60% [81]. In one
study, the overall survival at 18 months was 71% and disease free
survival was 34% [67]. In another study, the one, 2, and 5 year
survival rates were 78%, 57%, and 27% in patients with stage I
lung cancer [69,83]. Overall, it has been suggested that thermal
ablation is non-inferior to stereotactic radiation therapy for
stage I non-small cell lung cancer with similar survival rates out
to 5 years post therapy [111]. Smaller lesions (less than 2-3 cm)
are more likely to demonstrate complete necrosis following therapy
which is associated with a better long term survival
[67,69,82,108]. Successful ablation is also associated with tumor
location more than 3-10 mm away from a vessel, and peripheral
location of the tumor [81].There may be a synergistic effect
between chemotherapy and RF ablation- particularly for patients
with metastatic disease [69]. Tumor recurrence can be local (most
common)- 38% of cases (within the margin of the ablated site),
intrapulmonary (18% of cases), nodal (18%), mixed local and nodal
(6%), or distant metastases (21%) [73]. Larger tumors and patients
with higher staged disease are at a higher likelihood for
recurrence [73].
Complications include pleuritic chest pain and a fever (for up to
1 week) in nearly all patients [58]. Other complications include
self-limited hemoptysis (10%) [67] that can last 2-4 weeks [79],
hoarseness, and skin burn [79]. More serious complications include
pneumothorax (28% to 54% of cases and between 10-33% require a
chest tube [58,60,67,68,69]), cavitations (20%) [58],
intrapulmonary hemorrhage (6-11%) [58,67]. A pleural effusion can
be seen in 19-60% of patients within 24-48 hours following
treatment [60,67,81], but less than 5% require thoracentesis
[58]. Other complications include phrenic nerve injury,
abscess, and bronchopleural fistula [79]. Overall 30 day procedure
specific mortality has been reported to be 2.6% [69]. Rib
fractures can be seen as a late complication of RFA or MWA
treatment in up to 13.5% of patients [89]. The risk for a late rib
fracture is higher in patients that have had ablations performed
close to the chest wall (especially when the treatment zone
invovled the pleura) and may be related to local bone
damage/necrosis associated with the procedure [89]. Women are also
3 times more likely to develop a fracture compared to men [89].
For patients with an implanted cardiac device, cardiology
interogation of the device before and following the procedure is
recommended [79]. Pacemakers should be set for continuous pacing
prior to the ablation [79]. During the procedure, cardiac
defibrillators should be turned off and emergency defibrillator
equipment should be available [79].
Combination chemoembolization and RF ablation of lung tumors may
produce a synergistic effect with greater tumor control [90].
Radiofrequency ablation can also be used in combination with or
following chemotherapy [93].
Following successful ablation, lesions appear as low-attenuation
areas with complete lack of contrast enhancement consistent with
necrosis [60,73]. Partially ablated lesions show various
enhancement patterns [60]. Reactive hyperemia in the tissue
surrounding the ablated lesion represents an inflammatory reaction
to the thermal injury and this can be seen on studies performed
shortly after therapy [60]. A thin symmetric smooth rim of
peripheral enhancement of less than 5 mm width can be seen for up
to 6 months after ablation [73,81]. Irregular focal enhancement of
more than 15 HU compared with an unenhanced exam is considered
evidence of local tumor progression [73]. Following ablation, a
zone of surrounding ground glass attenuation is commonly seen and
typically resolves within 1 to 3 months [79]. Cavitation can be
seen in 24-31% of tumors within the first 3 months following RF
ablation and is more commonly seen in larger tumors [79]. RFA
treated lesions show a relatively slow rate of involution with a
40% decrease in size at 15 months after treatment [108]. The
apparent tumor size is variable within the first 6 months after RF
ablation and there can be an apparent increase in size within the
first 3 months after the procedure [79]. By 6 months, the size of
the ablation zone should be the same or smaller than the original
tumor [81]. RFA zones that demonstrate continued growth beyond 6
months are highly suggestive of residual malignancy (growth at 3
months is concerning, but requires followup) [79,81]. However,
other authors suggest that during the initial post ablation period
(<2 months) the area of dense opacification and surrounding GGO
should serve as the new baseline and any increase in size of the
lesion on followup should be considered local progression [108].
Risk factors for local recurrence following RF ablation include
tumor size larger than 2 cm and tumor contact with large blood
vessels [90]. The recurrence rate has been reported to be 5% for
tumors smaller than 2 cm, but 13% for those larger than 2 cm [90].
An increased risk for local recurrence is also associated with
KRAS mutant adenocarcinoma) compared to KRAS wild-type lesions
[104].
Other findings that suggest recurrence on CT include an area of
nodular enhancement that measures more than 10mm, enhacement
greater than noted at baseline, focal central enhancement,
peripheral nodular growth, change from ground-glass to solid
opacity, and new lymph node enlargement [81]. However, lymph node
enlargement is commonly seen after ablation and is likely reactive
(these nodes should decrease in size at 3 and 6 months) [81].
Followup imaging is required to determine if the lymph nodes
continue to increase in size over time [81].
Click
here for for PET findings after RFA
Other percutaneous interventions include microwave ablation (WMA)
[79], cryotherapy [65,79], laser ablation, or transpulmonary
chemoembolization [64].
MWA uses microwaves to cause friction between water molecules,
generating hyperthermia [108]. The microwave energy spectrum is
higher than that used for radiofrequency ablation and results in a
much broader deposition of energy (up to 2 cm surrounding the
antenna) and hence, a larger zone of active heating [79].
Additionally, MWA can be employed with the use of multiple probes
(as opposed to a single probe with RFA) which permits a larger
ablation zone [108]. Following MWA, lesion involution is similar
to that observed following RFA [108].
Cryoablation utilizes pressurized argon gas to generate sub-zero
temperatures and can cool to as low as -140? C which results in an
ice ball formation and tissue destruction [79,108]. When
temperatures are below -40?C, protein denaturation, cell rupture,
and ischemia occur [108]. Unlike heat-based ablation, cryoablation
dies not create GGO intraprocedurally; instead the ice ball is
used to estimate the ablated margin [108]. Most ablation protocols
call for three freeze-thaw cycles to achieve tissue necrosis
[108].
Compared to RFA and MWA, cryoablated lesions show faster
involution on CT [108].
PET Imaging in Bronchogenic Carcinoma
REFERENCES:
(2) J Thorac Imag 1995, 10: 99-105
(3) J Thorac Imag 1997; 12: 159-172
(4) Radiographics 1997; 17: 377-390
(5) AJR 1997; 169: 355-367
(6) AJR 1997; High-resolution CT findings of mucinous bronchioloalveolar carcinoma: a case of pseudopulmonary alveolar proteinosis. 168: 99-100 (No abstract available)
(7) J Thorac Imag 1995, 10: 96-98
(8) Radiology 1997; 202: 435-439
(9) Radiology 1997; 202: 441-446
(11) J Thorac Imag 1995; 10: 106-111
(12) RadioGraphics 1997; 17: 1345-1357
(13) RadioGraphics 1998; 18: 5-20
(15) J Nucl Med 1998; 39: 234-236
(19) Radiology 1998; 207: 431-437
(20) AJR 1998; Chartrand-Lefebvre C, et al. Association of small cell lung cancer and the anti-Hu paraneoplastic syndrome: Radiographic and CT findings. 170: 1513-1517
(21) J Nucl Med 1998; Higashi K, et al. Fluorine-18-FDG PET imaging is negative in bronchoalveolar cell carcinoma. 39: 1016-1020
(22) AJR 1998; Aquino SL, et al. Distinction of consolidative bronchoalveolar cell carcinoma from pneumonia: Do CT criteria work? 171: 359-363
(23) Radiology 1999; Maldonado RL. The CT angiogram sign. 210: 323-324 (No abstract available)
(24) Chest 1999; Mastin ST, et al. FDG SPECT in patients with lung masses. 115: 1012-1017
(25) J Nucl Med 1999; Coleman RE. PET in lung cancer. 40: 814-820
(26) J Thorac Imag 1999; Bepler G. Lung cancer epidemiology and genetics. 14: 228-234
(27) AJR 1999; Akira M, et al. High-resolution CT findings of diffuse bronchoalveolar carcinoma in 38 patients. 173: 1623-1629
(28) J Thorac Imag 2000; Franklin WA. Pathology of lung cancer. 15: 3-12
(29) AJR 2000; Patz EF, et al. Prognostic value of thoracic FDG PET imaging after treatment for non-small cell lung cancer. 174: 769-774
(30) Radiology 1996; Kaneko M, et al. Peripheral lung cancer: screening and detection with low-dose spiral CT. 201: 798-802
(31) Lancet 1999; Henschke C, et al. Early lung cancer action project: Overall design and findings from baseline screening. 354: 99-105
(32) Society of Thoracic Radiology Course Syllabus 2000; Swenson SJ. Lung cancer screening: Low dose CT. 32-36
(33) Radiology 1999; Kakinuma R, et al. Detection failures in spiral CT screening for lung cancer: analysis of CT findings. 212: 61-66
(34) Society of Thoracic Radiology Course Syllabus 2000; Ginsberg MS. Unusual presentations of lung cancer. 37-38
(35) Society of Thoracic Radiology Course Syllabus 2000; Jett JR. Treatment of unresectable lung cancer. 49-50
(36) Radiology 2000; Goo JM, et al. Pulmonary tuberculoma evaluated by means of FDG PET: Findings in 10 cases. 216: 117-121
(37) Radiographics 2000; Patz EF. Evaluation of focal pulmonary abnormalities with FDG PET. 20: 1182-1185 (No abstract available)
(38) N Engl J Med 2000; Pieterman RM, et al. Preoperative staging of non-small-cell lung cancer with positron-emission tomography. 343: 254-261
(39) AJR 2000; Boiselle PM, et al. Lung cancer detection in the 21st century: Potential contributions and challenges of emerging technologies. 175: 1215-1221 (No abstract available)
(40) Chest 2000; McCain TW, et al. The usefulness of positron emission tomography in evaluating patients for pulmonary malignancies. 118: 1610-1615
(41) Thorax 1998; Lowe VJ, Naunheim KS. Current role of positron emission tomography in thoracic oncology. 53: 703-712
(42) Radiographics 1999; Rosado de Christenson ML, et al. Thoracic carcinoids: Radiologic-Pathologic correlation. From the archives of the AFIP. 19: 707-736
(43) AJR 1998; Erasmus JJ, et al. Evaluation of primary pulmonary carcinoid tumors using FDG PET. 170: 1369-1373
(44) J Nucl Med 1999; Delbeke D. Oncological applications of FDG PET imaging: Brain tumors, colorectal cancer, lymphoma, and melanoma. 40: 591-603
(45) Ann Thorac Surg 1998; Lowe VJ, Naunheim KS. Positron emission tomography in lung cancer. 65: 1821-29
(46) J Nucl Med 2001; Avril N, et al. Glucose metabolism of breast cancer assessed by 18F-FDG PET: Histologic and immunohistochemical tissue analysis. 42: 9-16
(47) J Clin Oncol 1998; Lowe VJ, et al. Prospective investigation of positron emission tomography in lung nodules. 16: 1075-84
(48) Eur Respir J 1999; Bury T, et al. Value of FDG-PET in detecting residual or recurrent nonsmall cell lung cancer. 14: 1376-1380
(49) J Clin Oncol 2001; Kalff V, et al. Clinical impact of 18F fluorodeoxyglucose positron emission tomography in patients with non-small-cell lung cancer: A prospective study. 19: 111-118
(50) Chest 2001; Dunagan DP, et al. Staging by positron emission tomography predicts survival in patients with non-small cell lung cancer. 119: 333-339
(51) Chest 2001; Hauber HP, et al. Positron emission tomography in the staging of small-cell lung cancer. A preliminary study. 119: 950-954
(52) J Cancer Res Clin Oncol 2000; Ilknur AK, et al. Positron emission tomography with 2-[18F] fluoro-2-deoxy-D-glucose in oncology: Part II: The clinical value in detecting and staging primary tumours. 126: 560-574
(53) Thorax 2002; Mulshine JL, Smith RA. Lung cancer 2: screening and early diagnosis of lung cancer. 57: 1071-1078
(54) Radiology 2003; Suh RD, et al. Unresectable pulmonary malignancies: CT-guided percutaneous radiofrequency ablation- preliminary results. 229: 821-829
(55) AJR 2004; Oshiro Y, et al. CT findings of surgically resected large cell neuroendocrine carcinoma of the lung in 38 patients. 182: 87-91
(56) J Nucl Med 2003; Kamel EM, et al. Whole-body 18F-FDG PET improves the management of patients with small cell lung cancer. 44: 1911-1917
(57) Radiology 2004; Lee JM, et al. Percutaneous radiofrequency ablation for inoperable non-small cell lung cancer and metastases: preliminary report. 230: 125-134
(58) AJR 2004; Steinke K, et al. Percutaneous radiofrequency ablation of lung tumors with expandable needle electrodes: tips from preliminary experience. 183: 605-611
(59) AJR 2004; Belfiore G, et al. CT-guided radiofrequency ablation: a potential complementary therapy for patients with unresectable primary lung cancer- a preliminary report of 33 patients. 183: 1003-1011
(60) AJR 2004; Jin GY, et al. Primary and secondary lung malignancies treated with percutaneous radiofrequency ablation: evaluation with followup helical CT. 183: 1013-1020
(61) Radiographics 2004; Lee KS, et al. T1 non-small cell lung cancer: imaging and histopathologic findings and their prognostic implications. 24: 1617-1636
(62) AJR 2005; van Sonnenberg E, et al. Radiofrequency ablation of thoracic lesions: Part 2, initial clinical experience- technical considerations in 30 patients. 184: 381-390
(63) Chest 2004; Libby DM, et al. Managing the small pulmonary nodule discovered by CT. 125: 1522-1529
(64) Radiology 2005; Vogl TJ, et al. Treatment of unresectable lung metastases with transpulmonary chemoembolization: preliminary experience. 234: 917-922
(65) Radiology 2005; Wang H, et al. Thoracic masses treated with percutaneous cryotherapy: initial experience with more than 200 procedures. 235: 289-298
(66) Radiographics 2006; Chong S, et al. Neuroendocrine tumors of the lung: clinical, pathologic, and imaging findings. 26: 41-58
(67) Radiology 2006; de Baere T, et al. Midterm local efficacy and survival after radiofrequency ablation of lung tumors with minimum follow-up of 1 year: prospective evaluation. 240: 587-596
(68) Radiology 2006; Hiraki T, et al. Pneumothorax, pleural effusion, and chest tube placement after radiofrequency ablation of lung tumors: incidence and risk factors. 241: 275-283
(69) Radiology 2007; Simon CJ, et al. Pulmonary radiofrequency ablation: long-term safety and efficacy in 153 patients. 243: 268-275
(70) AJR 2007; Chong S, et al. Integrated PET/CT of pulmonary neuroendocrine tumors: diagnostic and prognostic implications. 188: 1223-1231
(71) J Nucl Med 2009; Zhao B, et al. Imaging surrogates of tumor response to therapy: anatomic and functional biomarkers. 50: 239-249
(72) Radiology 2009; Godoy MCB, Naidich DP. Subsolid pulmonary nodules and the spectrum of peripheral adenocarcinomas of the lung: recommended interim guidelines for assessment and management. 253: 606-622
(73) Radiology 2010; Beland MD, et al. Primary non-small cell lung cancer: review of frequency, location, and time of recurrence after radiofrequency ablation. 254: 301-307
(74) AJR 2010; Kligerman S, Abbott G. A radiologic review of the new TNM classification for lung cancer. 194: 562-573
(75) Radiographics 2010; UyBico SJ, et al. Lung cancer staging essentials: the new TNM staging system and potential imaging pitfalls. 30: 1163-1181
(76) J Nucl Med 2011; de Langen AJ, et al. Monitoring response to antiangiogenic therapy in non-small cell lung cancer using imaging markers derived from PET and dynamic contrast-enhanced MRI. 52: 48-55
(77) Radiographics 2011; Nair A, et al. Revisions to the TNM staging of non-small cell lung cancer: rationale, clinicoradiologic implications, and persistent limitations. 31: 215-238
(78) AJR 2011; Kligerman S, White C. Epidemiology of lung cancer
in women: risk factors, survival, and screening. 196: 287-295
(79) Radiology 2011; Dupuy DE. Image-guided thermal ablation of
lung malignancies. 260: 633-655
(80) AJR 2012; Nishino M, et al. Personalized tumor response
assessment in the era of molecular medicine: cancer-specific and
therapy-specific response criteria to complement pitfalls of
RECIST. 198: 737-745
(81) Radiographics 2012; Abtin F, et al. Radiofrequency ablation
of lung tumors: imaging features of the post-ablation zone. 32:
947-969
(82) J Nucl Med 2010; Singnurkar A, et al 18F-FDG
PET/CT for the prediction and detection of local recurrence after
radiofrequency ablation of malignant lung lesions. 51: 1833-1840
(83) AJR 2011; Yoo DC, et al. Radiofrequency ablation of
medically inoperable stage IA non-small cell lung cancer: are
early posttreatment PET findings predictive of treatment outcome?
197: 334-340
(84) Radiology 2012; Aoki T, et al. Adenocarcinomas with
predominantly ground-glass opacity: correlation of morphology and
molecular biomarkers. 264: 590-596
(85) Radiology 2012; Lee HY, et al. Solitary pulmonmary nodular
lung adenocarcinoma: correlation of histopathologic scoring and
patient survival with imaging biomarkers. 264: 884-893
(86) Radiology 2013; Austin JHM, et al. Radiologic implications
of the 2011 classification of adenocarcinoma of the lung. 266:
62-71
(87) Electronic reference: American Society of Clinical Oncology;
Brambilla E.
http://www.asco.org/ASCOv2/Home/Education%20%26%20Training/Educational%20Book/PDF%20Files/2011/zds00111000279.PDF
(88) Radiology 2013; Naidich DP, et al. Recommendations for the
management of subsolid pulmonary nodules detected at CT: a
statement from the Fleischner Society. 266: 304-317
(89) Radiology 2013; Alexander ES, et al. Rib fractures after
percutaneous radiofrequency and microwave ablation of lung tumors:
incidence and relevance. 266: 971-978
(90) Radiology 2013; Gadaleta CD, et al. Unresectable lug
malignancy: combination therapy with segmental pulmonary arterial
chemoembolization with drug-eluting microspheres and
radiofrequency ablation in 17 patients. 267: 627-637
(91) Radiology 2013; Lee SM, et al. Invasive pulmonary
adenocarcinomas versus preinvasive lesions appearing as
ground-glass nodules: differentiation by using CT features. 268:
265-273
(92) Radiographics 2013; Benson REC, et al. Spectrum of pulmonary
neuroendocrine proliferations and neoplasms. 33: 1631-1649
(93) AJR 2013; Li X, et al. Percutaneous CT-guided radiofrequency
ablation as supplemental therapy after systemic chemotherapy for
selected advanced non-small cell lung cancers. 201: 1362-1367
(94) Radiology 2014; Nishino M, et al. State of the art: response
assessment in lung cancer in the era of genomic medicine. 271:
6-27
(95) J Nucl Med 2014; van Gool MH, et al. Timing of metabolic
response monitoring during erlotinib treatment in non-small cell
lung cancer. 55: 1081-1086
(96) Radiographics 2014; Chou SHS, et al. Thoracic diseases
associated with HIV infection in the era of anti-retroviral
therapy: clinical and imaging findings. 34: 895-911
(97) Radiographics 2014; Carter BW, et al. Small cell lung
carcinoma: staging, imaging, and treatment considerations. 34:
1707-1721
(98) Radiographics 2015; Tirumani SH, et al. Anti-VEGF molecular targeted therapies in common solid malignancies: comprehensive update for radiologists. 35: 455-474
(99) AJR 2015; Howard SA, et al Beyond the vascular endothelial
growth factor axis: update on the role of imaging in
nonantiangiogenic molecular targeted therapy. 204: 919-932
(100) Radiology 2015; Cook GJR, et al.
Non-small cell lung cancer treated with Erlotinib: heterogeneity
of 18F FDG uptake at PET-
association with treatment response and prognosis. 276: 883-893
(101) Radiology 2016; Ryu JS, et al. Minimal
pleural effusion in small cell lung cancer: proportion,
mechanisms, and prognostic effect. 278: 593-600
(102) J Am Coll Radiol 2013; Gill RR, et al.
Controveries in lung cancer screening.
(103) Radiology 2016; Liu Y, et al. CT
features associated with epidermal growth factor receptor
mutation status in patients with lung adenocarcinoma. 280:
271-280
(104) Radiology 2017; Ziv E, et al. Lung
adenocarcinoma: predictive value of KRAS mutation status in
assessing local recurrence in patients undergoing image-guided
ablation. 282: 251-258
(105) J Nucl Med 2017; Yip SSF, et al.
Associations between somatic mutations and metabolic imaging
phenotypes in non-small cell lung cancer. 58: 569-576
(106) Clin Cancer Res 2008; Riley GJ, et al.
Frequency and distinctive spectrum of KRAS mutations in never
smokers with lung adenocarcinoma. 14: 5731-34
(107) Radiology 2017; Cox VL, et al Cancer
genomics and important mutations: a contemporary guide for body
imagers. 283: 314-340
(108) AJR 2017; Mouli SK, et al. The role of
percutaneous image-guided thermal ablation for the treatment of
pulmonary malignancies. 209: 740-751
(109) J Nucl Med 2017; Fledelius J, et al.
18F-FDG PET/CT for very early response evaluation predicts CT
response in erlotinib-treated non-small cell lung cancer
patients: a comparison of assessment methods. 58: 1931-1937
(110) AJR 2018; Chang S, et al. Adverse
prognostic CT findings for patients with advanced lung
adenocarcinoma receiving first-line epidermal growth factor
receptor-tyrosine kinase inhibitor therapy. 210: 43-51
(111) Radiology 2018; Uhlig J, et al.
Survival rates after thermal ablation versus sterotactic
radiation therapy for stage I non-small cell lung cancer: a
national cancer database study. 289: 862-870
(112) AJR 2019; Chen L, et al. A
radiologists guide to the changing treatment paradigm of advanced
non-small cell lung cancer: the ASCO 2018 molecular testing
guidelines and target therapies. 213: 1047-1058
(113) AJR 2020; Shao X, et al. ROle of PET/CT in management of
early adenocarcinoma. 214: 437-445
(114) J Nucl Med 2020; Niemeiher AL, et al. Imaging responses to
immunotherapy with novel PET tracers. 61: 641-642
(115) J Nucl Med 2020; Castello A, et al. Hyperprogressive
disease in patients with non-small cell lung cancer treated with
checkpoint inhibitors: the role of 18F-FDG PET/CT. 61:
821-826
(116) J Nucl Med 2020; Comparison between 18F-FDG
PET-based and CT-based criteria in non-small cell lung cancer
patients treated with Nivolumab. 61: 990-998
(117) Radiology 2020; Kuriyama K, Yanagawa M. CT diagnosis of
lung adenocarcinoma: radiologic-pathologic correlation and growth
rate. 297: 199-200