Patients with increasing lymphoma tumor metabolism on PET/CT scans prior to CAR-T therapy have an increased risk of death, according to a group at the Mayo Clinic in Rochester, MN.
A team led by radiation oncologist William Breen, MD, developed what they described as a simple score for predicting outcomes in patients based on two metrics observed on F-18 FDG-PET/CT scans prior to patients beginning CAR-T therapy. The tool could help tailor patient selection, they suggested.
"As CAR-T therapy utilization and indications increase, methods of prognostic differentiation using noninvasive methods such as PET/CT are needed to identify which patients are at the highest risk of [progressive disease]," the group wrote, in a study published August 18 in the Blood Cancer Journal.
CAR-T (chimeric antigen receptor T-cell) therapy is among the latest treatments for blood cancer. The immunotherapy treatment involves extracting and genetically modifying a patient's T-cells to boost their ability to destroy cancer cells, and then reintroducing those cells.
At approximately six weeks prior to therapy, doctors remove the T-cells to modify them. This is the pre-leukapheresis, or "pre-leuk" stage, the authors noted. Then, at one week prior to therapy, patients undergo chemotherapy to clear space for the engineered CAR-T cells (the pre-lymphodepletion chemotherapy, or the "pre-LD" stage).
In practice, F-18 FDG-PET/CT scans are used to evaluate the extent of disease at both of these time points, yet it is unknown whether changes in disease between the scans are prognostic, the authors noted.
To elucidate the role of PET/CT in this regard, the group analyzed data from 69 patients with aggressive relapsed or refractory B-cell non-Hodgkin lymphoma who received axicabtagene ciloleucel (Yescarta) between January 2018 and July 2020. Pre-leuk and pre-LD PET/CT scans were performed a median of 46 days and seven days prior to CAR-T infusion. At the last follow-up, 46 patients (67%) had experienced progressive disease and 30 patients (43%) had died.
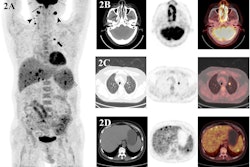
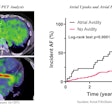
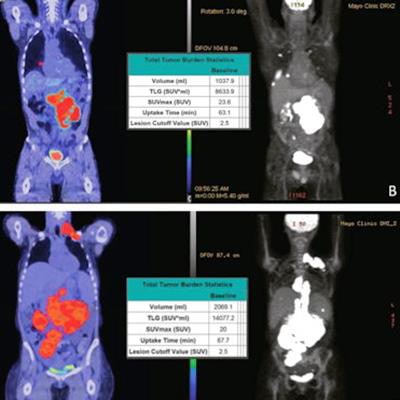
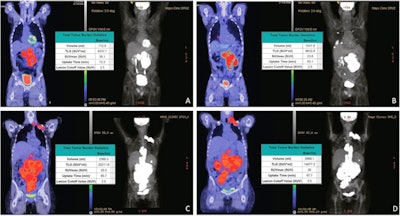 Pre-leukapheresis (pre-leuk) and pre-lymphodepletion chemotherapy (pre-LD) coronal fused PET/CT (left) and maximum projection image (right) PET/CT imaging and total tumor volumetric analysis for two patients. The first patient A, B had both increasing extranodal metabolic tumor volume (MTV) and increasing total lesion glycolysis (TLG) of the largest lesion (risk score 2) from pre-leuk A to pre-LD B, and experienced progressive disease and death after CAR-T infusion. The second patient C, D had neither risk factor (risk score 0), is alive and without progression at last follow-up, despite having higher MTV and TLG at each individual time point compared to the first patient. Image and caption courtesy of the Blood Cancer Journal through CC BY 4.0.
Pre-leukapheresis (pre-leuk) and pre-lymphodepletion chemotherapy (pre-LD) coronal fused PET/CT (left) and maximum projection image (right) PET/CT imaging and total tumor volumetric analysis for two patients. The first patient A, B had both increasing extranodal metabolic tumor volume (MTV) and increasing total lesion glycolysis (TLG) of the largest lesion (risk score 2) from pre-leuk A to pre-LD B, and experienced progressive disease and death after CAR-T infusion. The second patient C, D had neither risk factor (risk score 0), is alive and without progression at last follow-up, despite having higher MTV and TLG at each individual time point compared to the first patient. Image and caption courtesy of the Blood Cancer Journal through CC BY 4.0.To assess the PET/CT scans for associations with outcomes, the researchers used metabolic tumor volume (MTV) and total lesion glycolysis (TLG) as metrics, as these metabolic data have shown prognostic value in lymphoma patients undergoing other treatments.
According to the findings, neither of the single time-point PET/CT scans predicted risk, yet increases in MTV and TLG between the pre-leuk to pre-LD scans did, the researchers wrote. Specifically, increasing extranodal MTV and increasing TLG of the largest lesions were the strongest predictors of death, with an area under the curve of 0.74.
"Patients with increasing metabolic characteristics from pre-leuk to pre-LD PET/CT have increased risk of progression and death," the group stated.
Ultimately, CAR-T therapy has demonstrated promising outcomes for patients with aggressive lymphoma and provides an opportunity for long-term remission. However, given the considerable costs and rates of disease progression following the treatment, advances in risk stratification are needed, the researchers noted.
Thus, the "two variable risk score" developed in this study may be used to stratify prognosis prior to CAR-T infusion and may inform clinicians when considering risk-adapted strategies, they added.
"This study provides clinically useful information to help inform prognosis and potentially identify patients who may benefit from treatment intensification," the group concluded.
The full article is available here.