HIV:
Also refer to the specific disorder that you are interested in for a detailed description of the findings associated with HIV.Clinical:
Patients with HIV infection are at increased risk for many pulmonary infections including encapsulated bacteria [10] (such as Strep pneumoniae and H. influenza), PCP, Mycobacterial disease- both TB and MAC, and CMV. Almost 70% of AIDS patients suffer a respiratory episode during the course of their illness [7].The patients degree of immunosuppression (CD4 count), epidemologic factors, geography, and use of antipneumocystis prophylaxis will usually determine the type of infection that they will develop [5,11]. Normal values for CD4 lymphocytes (T-helper lymphocytes) range between 800-1000 cells/uL. At CD4 levels above 500 cells/uL, HIV patients are at risk for infections from more virulent organisms such as bacterial pneumonias or TB [7]. Between 200 and 499 cells/uL, HIV patients may experience recurrent bacterial pneumonias or TB [7,11]. When the CD4 count drops below 200 cells/uL, PCP infection is common [7]. Patients are also at increased risk for disseminated TB infection [11]. (Note: A CD4 count of 200 cells/uL indicates the onset of clinical AIDS in the HIV positive patient even in the absence of opportunistic infection [7]). In very late stages of HIV (CD4 counts below 50 cells/uL) patients are at high risk for MAC, CMV, and fungal infection as well as AIDS-related lymphoma and Kaposi's sarcoma [11]. Patients with CD4 counts below 50 cells/uL have a median survival between 12 to 18 months.
New antiretroviral regimens that use combinations of nucleoside analogues with or without protease inhibitors offer the possibility of longer disease free survival in HIV patients. With this prolonged survival, changes in the types of infections experienced by HIV patients may be anticipated.
Immune reconstitution inflammatory syndrome/Immune restoration syndrome/Immune reconstitution inflammatory syndrome:
Highly active antiretroviral therapy (HAART) has also be
associated with an immune reconstitution syndrome (IRS) [12,13].
The syndrome typically develops within 1to 3 months after
initiation of HAART and coincides with rising CD4 counts (although
the onset may occur years after HAART) [13,14,17]. In this
syndrome, there is an a temporary worsening or unmasking of
infectious or malignant disease following initiation of HAART
[12,19]. The condition is felt to be due to restoration of the
immune system that incites an inflammatory reaction against
infectious and non-infectious antigens (TB,MAI, cryptococcus, CMV,
or HSV) [12,13,15]. The response is exaggerated because of a lack
of homeostatic regulation [15]. It is characterized by a
deterioration in clinical condition despite improvement in HIV RNA
level and increasing CD4 count [12]. It is estimated that
10-25% of patients who start HAART experience immune
reconstitution syndrome [12,14,17]. Patients at greatest risk for
developing IRS have a low CD4 count (<50 cells/uL) prior to
initiation of HAART, a high viral load (>100,000 copies/mL),
and a rapid decrease in plasma HIV RNA during the first 3 months
of therapy [14]. Also- the initiation of HAART soon after the
diagnosis of an opportunistic infection has also been associated
with an increased risk for IRS [14]. Up to 30% of patients with a
known opportunistic infection experience this clinical
deterioration following the initiation of HAART (despite having no
progression of their HIV disease and no new secondary infection)
[17,18].
The process is also most likely to develop in patients with
untreated or partially treated TB or nontuberculous mycobacterial
infection [17] (tuberculous-IRS is the common form consisting of
either a paradoxical worsening or unmasking of latent infection
with fever, recurrent TB symptoms, consolidation, centrilobular
nodules,and adenopathy within the first two months of HAART [18]).
Neoplasms such as Kaposi sarcoma have also shown transient
worsening related to initiation of HAART [17]. Lymphadenopathy is
a common imaging feature of IRS, commonly with low attenuation
centers due to necrosis of suppuration [19]. Sarcoidosis has been
described as a late manifestation of immune reconstitution
syndrome- however, the majority of HIV patients with sarcoid have
CD4 counts > 200 and present months to years after initiation
of HAART [17].
Patients present with fever, abdominal pain, night sweats, cough,
and enlarged lymph nodes [13]. Findings on CT include enlarging
lymph nodes (73% of patients) that appear necrotic in up to 88% of
cases [13]. Other findings include new or worsening lung nodules
with a diameter of less than 3 mm (55% of patients),
microabscesses in the liver and spleen (18%), small pleural
effusions (18%), and small volume ascites (18%) [13]. For the
diagnosis of IRS to be made, the patients condition must improve
when HAART is initiated and then become worse, and the viral load
should decrease as the CD4 count increases [18].
Bacterial Infection:
i) Pyogenic Airway Disease:
Airway disease (acute bronchitis) is the most frequent disorder of the lower respiratory tract and it occurs at all levels of immunosuppression [3]. Patients typically present with cough productive of purulent sputum, fever, and dyspnea [7]. Small airways disease may not be detected on plain film radiographs and a high resolution CT scan should be obtained if the diagnosis is being considered [7]. Even then, airway infection may not be evident on CT scan. Findings indicative of pyogenic airway infection include dilatation and thickening of the bronchial walls due to bronchial or peribronchial inflammation [7]. Findings of bronchiolitis may also be identified such as small (1-3 mm), ill-defined centrilobular "Y" or "V" shaped densities (due to impacted inflammatory material within bronchioles) [1,7]. The lower lobes are usually affected [7]. Focal areas of air trapping may be present and will be accentuated by images obtained at end-expiration (this will aid in distinguishing heterogeneous parenchymal attenuation due to air trapping from ground-glass attenuation secondary to PCP [7]).ii) Bacterial Pneumonia:
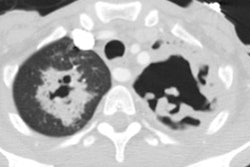
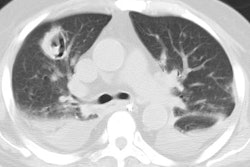
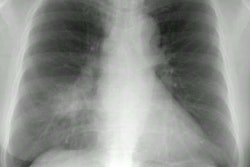
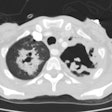
Bacterial pneumonia is 10-25 times more common in HIV patients than in the general population [18]. Pyogenic infections (bacterial pneumonias and bronchitis) are more common than PCP in HIV patients with both early and severe immunocompromise (this may be the result of routine use of PCP prophylaxis). Bacterial pneumonias occur in 5 to 30% of HIV patients, most commonly before their first AIDS-defining illness (CD4 counts between 200-500 cells/uL). The rate at which HIV patients develop pneumonia is inversely related to their CD4 count [3] (ie: the risk of bacterial infection increases steadily with decreasing CD4 lymphocyte counts [10]). More than two episodes of bacterial pneumonia within one year is AIDS-defining in an HIV infected patient, regardless of their CD4 count [9]. Bacterial pneumonias also occur with the highest incidence in HIV positive intravenous drug abusers as compared with other HIV-positive groups [7,10].The most common bacterial pneumonias found in HIV patients are with the encapsulated organisms S. pneumoniae and H. influenzae (second most common organism) [2,3,10,16,18]. The incidence of pneumococcal pneumonia is 5 to 18 times that of the general population and the development of pneumoncoccal septicemia is 100 times greater [10,18]. Other causes of pneumonia include S. aureus (associated with IV drug use and tricuspid endocarditis), Legionella (40 fold higher incidence in HIV patients), and Rhodocossus equi (an acid fast coccobacillus that can manifest in advanced HIV patients with an indolent course of fever, cough, dyspnea, and upper lobe consolidation or cavitary masses mimicking TB) [18].
The presenting features of community acquired pneumonia in HIV patients is similar to that of non-HIV individuals, but they have a higher frequency of bacteremia, a higher frequency of multilobar infection, and a tendency to relapse [3,9]. Bacterial pneumonias in AIDS patients tend to progress more rapidly, and are more often complicated by cavitation and abscess formation [10]. Pneumococcal vaccine is recommended for all HIV infected persons [5]. Hospital acquired pneumonias (especially with enteric gram negative bacilli and staphylococci) are common in HIV patients with advanced disease and are frequently a terminal event [3]. Radiographically, focal infiltrates are the most common finding in HIV patients with bacterial pneumonia.
Uncommon nontuberculous mycobacterial infections (such as Mycobacterium
xenopi [from contaminated hospital water supplies] and Mycobacterium
genavense) can also been seen in HIV patients [8]. Chest
radiographs typically reveal an interstitial infiltrate without
associated adenopathy, although the film can be normal in up to
14% of cases [8].
iii) Mycobactrium
tuberculosis
Fungal Infection:
Fungal infections in HIV patients are unusual, even with advanced levels of immune compromise. Cryptococcus is generally considered the most common fungal pneumonia in HIV patients [6,16]. However, this may be controversial- McGuinness states that Aspergillosis is the most common pulmonary infection [7].Cryptococcal pneumonia accounts for between 2-15% of AIDS related pneumonias [4]. Patients with cryptococcal pneumonia usually have CD4 counts below 100 and frequently have disseminated disease and often have meningitis as well- a concomitant meningitis is found in up to 86% of cases, although it commonly clinically silent. Conversely, in HIV patients that present with CNS cryptococcal infection, clinically silent pulmonary disease is found in just over 25% of cases. Radiographic findings include a coarse asymmetric nodular or interstitial infiltrate or alveolar consolidations, however, the CXR can be normal (14% of cases in a small series) [4].
Click here for more on Cryptococcal infection in HIV patients
Aspergillus infection is not commonly seen in HIV because the main host defense against aspergillus is the neutrophil [9]. Aspergillosis, however, is the most common fungal pathogen to cause airway disease in HIV patients. Infection typically occurs late in the course of HIV infection, often in association with other risk factors such as steroid therapy, granulocytopenia, or IV drug use [1]. Marijuana smoking is also a risk factor for aspergillus infection because marijuana contains the organism which is ubiquitous in soil and water [9].
Click here for more on Aspergillis infection in HIV patients
Histoplasmosis and coccidiomycosis can also both occur in HIV patients [3].
Adenopathy:
Persistent generalized adenopathy is defined as palpable lymphadenopathy larger than 1 cm, involving two or more non contiguous nodal groups lasting longer than 3 months [19]. Lymphoid hyperplasia is the most common cause of generalize adenopathy in patients with HIV infection [19]. Low CD4 counts correlate with increased incidence of lymphadenopathy [19]. The most commonly involved site is the head and neck and it is commonly accompanied by nasopharyngeal lymphatic tissue hypertrophy and cystic changes in the parotid gland (proliferation of the intraglandular lymphoid tissue obstruction the epithelium resulting in cystic enlargment [19].Neoplasms:
HIV patients are also at risk for pulmonary neoplastic disease (Kaposis sarcoma and bronchogenic carcinoma) and lymphoproliferative disorders such as lymphocytic interstitial pneumonitis, and AIDS related lymphoma.REFERENCES:
(1) AJR 1997; 168: 67-77
(2) ACR Syllabi #40:p.120-21;122-125
(3) Annu Rev Med 1996; 47: 117-26
(4) Clin Radiol 1995; 50: 756-760
(5) Curr Opin Pulm Med 1997; 3: 151-158
(6) Semin Roentgenology, Jan 96, p67-75
(7) Radiologic Clinics of North America 1997; McGuinness G. Changing trends in the pulmonary manifestations of AIDS. 35 (5): 1029-1082
(8) Chest 1998; El-solh AA, et al. Clinical and radiographic manifestations of uncommon pulmonary nontuberculous mycobacterial disease in AIDS patients. 114: 138-145
(9) J Thorac Imaging 1998; Haramati LB, et al. Approach to the diagnosis of pulmonary disease in patients infected with the human immunodeficiency virus. 13: 247-260
(10) AJR 2003; Brecher CW, et al. CT and radiography of bacterial respiratory infections in AIDS patients. 180: 1203-1209
(11) AJR 2005; Marchiori E, et al. Pulmonary disease in patients with AIDS: high-resolution CT and pathologic findings. 184: 757-764
(12) AJR 2007; Godoy MCB, et al. Imaging features of pulmonary kaposi sarcoma-associated immune reconstruction syndrome. 189: 956-965
(13) Radiographics 2009; Johnson PT, et al. Nonvascular mesenteric disease: utility of multidetector CT with 3D volume rendering. 12: 721-740
(14) Radiology 2009; Chen KC, et al. Immune reconstitution
syndrome. 252: 924-928
(15) AJR 2011; Davison JM, et al. FDG PET/CT in patients with
HIV. 197: 284-294
(16) AJR 2012; Lichtenberger JP, et al. What a differetial a
virus makes: a practical approach to thoracic imaging findings in
the context of HIV infection- Part I, pulmonary findings. 198:
1295-1304
(17) AJR 2012; Lichtenberger JP, et al. What a differetial a
virus makes: a practical approach to thoracic imaging findings in
the context of HIV infection- Part 2, extrapulmonary pulmonary
findings, chronic lung disease, and immune reconstitution
syndrome. 198: 1305-1312
(18) Radiographics 2014; Chou SHS, et al. Thoracic diseases
associated with HIV infection in the era of anti-retroviral
therapy: clinical and imaging findings. 34: 895-911
(19) AJR 2021; Glushko T, et al. HIV lymphadenopathy:
differential diagnosis and important imaging features. 216:
526-533